1 PITAVASTATIN
2 ATORVASTATIN
3
4
5

1 PITAVASTATIN







......................................
2 ATORVASTATIN







.....................
3.
..........................................

............................
CITRUS AND STATINS
..........................................

2 ATORVASTATIN
3
4
5

1 PITAVASTATIN

PITAVASTATIN, LIVALO, Itavastatin calcium, Nisvastatin, NKS-104, NK-104,
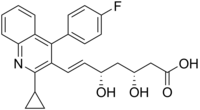
(3R, 5S) -7 – [2-Cyclopropyl-4-(4-fluorophenyl) quinolin-3-yl] -3,5-dihydroxy-6 (E)-heptenoic acid calcium salt (2:1)
CAS REGISTRY NUMBER
147526-32-7 CA SALT, 147511-69-1 (free acid), 141750-63-2 (lactone), 192565-91-6 (monoK salt)
rotation is +
alpha(D20) +6.8° (c 1.74, CHCl3)
ALSO
Bioorganic and Medicinal Chemistry Letters, 1999 , VOL 9, 20 pg. 2977 – 2982…….alpha(D20) +23.1° (c 1.0, acn/water(1:))
Helvetica Chimica Acta, 2007 , vol. 90, 6 pg. 1069 – 1081…alpha(D20) +22.9° (c 1.0, acn/water)
(3R,5S,6E)-7-[2-cyclopropyl-4-(4-fluorophenyl)quinolin-3-yl]-3,5-dihydroxyhept-6-enoic acid
Pitavastatin a lipid-lowering agent that belongs to the statin class of medications for treatment of dyslipidemia. It is also used for primary and secondary prevention of cardiovascular disease. FDA approved in Aug 3, 2009.
2-C25-H23-FN-O4.Ca, 881.01
Nissan Chemical (Originator), Kowa (Licensee), Novartis (Licensee), Recordati (Licensee), Sankyo (Licensee)

Lipoprotein Disorders, Treatment of, METABOLIC DRUGS, APOA1 Expression Enhancers, HMG-CoA Reductase Inhibitors, SPP1 (Osteopontin) Expression Inhibitors
Launched-2003
Statin drugs are currently the most therapeutically effective drugs available for reducing the level of Low density lipoprotein (LDL) in the blood stream of a patient at risk for cardiovascular disease. A high level of LDL in the
bloodstream has been linked to the formationof coronary lesions which obstruct the flow of blood and can rupture and promote thrombosis. It is well known that inhibitors against HMG CoA reductase which is rate limiting enzyme for cholesterol biosynthesis have been clinically proved to be potentially useful anti-hyperlipoproteinemic agents
and they are considered very effective curative and preventive for coronary artery sclerosis or atherosclerosis .
Pitavastatin calcium was discovered by Nissan Chemical Industries Limited Japan and developedfurther by Kowa Pharmaceuticals Tokyo Japan is a novel member of the medication class of statins.
bloodstream has been linked to the formationof coronary lesions which obstruct the flow of blood and can rupture and promote thrombosis. It is well known that inhibitors against HMG CoA reductase which is rate limiting enzyme for cholesterol biosynthesis have been clinically proved to be potentially useful anti-hyperlipoproteinemic agents
and they are considered very effective curative and preventive for coronary artery sclerosis or atherosclerosis .
Pitavastatin calcium was discovered by Nissan Chemical Industries Limited Japan and developedfurther by Kowa Pharmaceuticals Tokyo Japan is a novel member of the medication class of statins.
LIVALO (pitavastatin) is an inhibitor of HMG-CoA reductase. It is a synthetic lipid-lowering agent for oral administration.
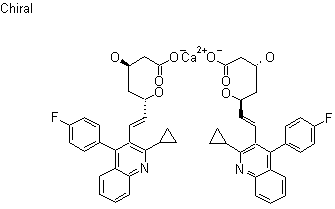
The chemical name for pitavastatin is (+)monocalcium bis{(3R, 5S, 6E)-7-[2-cyclopropyl-4-(4-fluorophenyl)-3-quinolyl]-3,5dihydroxy-6-heptenoate}. The structural formula is:
 |
The empirical formula for pitavastatin is C50H46CaF2N2O8 and the molecular weight is 880.98. Pitavastatin is odorless and occurs as white to pale-yellow powder. It is freely soluble in pyridine, chloroform, dilute hydrochloric acid, and tetrahydrofuran, soluble in ethylene glycol, sparingly soluble in octanol, slightly soluble in methanol, very slightly soluble in water or ethanol, and practically insoluble in acetonitrile or diethyl ether. Pitavastatin is hygroscopic and slightly unstable in light.
Each film-coated tablet of LIVALO contains 1.045 mg, 2.09 mg, or 4.18 mg of pitavastatin calcium, which is equivalent to 1 mg, 2 mg, or 4 mg, respectively of free base and the following inactive ingredients: lactose monohydrate, low substituted hydroxypropylcellulose, hypromellose, magnesium aluminometasilicate, magnesium stearate, and film coating containing the following inactive ingredients: hypromellose, titanium dioxide, triethyl citrate, and colloidal anhydrous silica.
Pitavastatin (usually as a calcium salt) is a member of the blood cholesterol loweringmedication class of statins,[1] marketed in the United States under the trade nameLivalo. Like other statins, it is an inhibitor of HMG-CoA reductase, the enzyme that catalyses the first step of cholesterol synthesis. It has been available in Japan since 2003, and is being marketed under licence in South Korea and in India.[2] It is likely that pitavastatin will be approved for use in hypercholesterolaemia (elevated levels of cholesterol in the blood) and for the prevention of cardiovascular disease outside South and Southeast Asia as well.[3] In the US, it received FDA approval in 2009.[4]
Pitavastatin is used to lower serum levels of total cholesterol, LDL-C, apolipoprotein B, and triglycerides, and raise levels of HDL-C for the treatment of dyslipidemia.
Like the other statins, pitavastatin is indicated for hypercholesterolaemia (elevated cholesterol) and for the prevention of cardiovascular disease. A 2009 study showed that pitavastatin increased HDL cholesterol (24.6%), especially in patients with HDL lower than 40 mg/dl, in addition to greatly reducing LDL cholesterol (–31.3%).[5] As a consequence, pitavastatin is most likely to be appropriate for patients with metabolic syndrome with high LDL, low HDL and diabetes mellitus.
Common statin-related side effects (headaches, stomach upset, abnormal liver function tests and muscle cramps) were similar to other statins. However, pitavastatin seems to lead to fewer muscle side effects than certain statins that are lipid-soluble, as a result of the fact that pitavastatin is water-soluble (as is pravastatin, for example).[6] One study found that coenzyme Q10 was not reduced as much as with certain other statins (though this is unlikely given the inherent chemistry of the HMG-CoA reductase pathway that all statin drugs inhibit).[3][7]
Hyperuricemia or increased levels of serum uric acid have been reported with pitavastatin.[8]
Most statins are metabolised in part by one or more hepatic cytochrome P450enzymes, leading to an increased potential for drug interactions and problems with certain foods (such as grapefruit juice). Pitavastatin appears to be a substrate ofCYP2C9, and not CYP3A4 (which is a common source of interactions in other statins). As a result, pitavastatin is less likely to interact with drugs that are metabolized via CYP3A4, which might be important for elderly patients who need to take multiple medicines.[3]
Pitavastatin (previously known as itavastatin, itabavastin, nisvastatin, NK-104 or NKS-104) was discovered in Japan by Nissan Chemical Industries and developed further byKowa Pharmaceuticals, Tokyo.[3] Pitavastatin was approved for use in the United States by the FDA on 08/03/2009 under the trade name Livalo. Pitavastatin has been also approved by the Medicines and Healthcare products Regulatory Agency (MHRA) in UK on 17 August 2010.
- Kajinami, K; Takekoshi, N; Saito, Y (2003). “Pitavastatin: efficacy and safety profiles of a novel synthetic HMG-CoA reductase inhibitor”.Cardiovascular drug reviews 21 (3): 199–215. PMID 12931254. edit
- Zydus Cadila launches pitavastatin in India
- Mukhtar, R. Y. A.; Reid, J.; Reckless, J. P. D. (2005). “Pitavastatin”. International Journal of Clinical Practice 59 (2): 239–252.doi:10.1111/j.1742-1241.2005.00461.x. PMID 15854203. edit
- The Seventh Statin; Pitavastatin
- http://www.ncbi.nlm.nih.gov/pubmed/19907105
- ScienceDaily (11 May 2013). “Alternative Cholesterol-Lowering Drug for Patients Who Can’t Tolerate Statins”. ScienceDaily.
- Clin Pharmacol Ther. 2008 May;83(5):731-9. Epub 2007 Oct 24. Comparison of effects of pitavastatin and atorvastatin on plasma coenzyme Q10 in heterozygous familial hypercholesterolemia: results from a crossover study. Kawashiri MA, Nohara A, Tada H, Mori M, Tsuchida M, Katsuda S, Inazu A, Kobayashi J, Koizumi J, Mabuchi H, Yamagishi M.
- Ogata, N.; Fujimori, S.; Oka, Y.; Kaneko, K. (2010). “Effects of Three Strong Statins (Atorvastatin, Pitavastatin, and Rosuvastatin) on Serum Uric Acid Levels in Dyslipidemic Patients”. Nucleosides, Nucleotides and Nucleic Acids 29 (4–6): 321.doi:10.1080/15257771003741323. edit

Country
|
Patent Number
|
Approved
|
Expires (estimated)
|
|---|---|---|---|
| United States | 7,022,713 | 2009-08-03 | 2024-02-19 |
| United States | 6,465,477 | 2009-08-03 | 2016-12-20 |
| United States | 5,856,336 | 2009-08-03 | 2016-01-05 |
| United States | 5,854,259 | 2009-08-03 | 2015-12-29 |
| United States | 5,753,675 | 2009-08-03 | 2015-05-19 |
JP 1993310700, JP 1994025092
Tetrahedron Lett 1993, 34, 51, 8263-6.
Bioorg Med Chem2001, 9, (10): 2727
Drugs Fut1998, 23, (8) :847-859
Bull Chem Soc Jpn1995, 68, (1) :364-72
Tetrahedron Asymmetry1993, 4, (2) :201-4
Tetrahedron Lett1993, 34, (51) :8267-70
A Endo; J. Med. Chem. 1985, 28, 401.
AM Gotta; LC Smith; IXth International Symp. Drugs Affecting Lipid Metabolism, 1986, 30- 31
Y Fujikawa, Nissan Chemical Industries Ltd, EP304063 (A3), 1989.
S Ahmed; CS Madsen; PD Stein; J. Med. Chem. 2008, 51, 2722-2733.
K Turner; Org. Process. Res. Dev, 2004, 8, 823-833.
Z Casar; M Steinbocher; J Kosmrlj; J. Org. Chem. 2010, 75(19), 6681-6684.
KL Baumann; Tetrahedron Letters, 1992, 33, 2283-2284.
N Miyachi; Y Yanagawa; H Iwasaki; Tetrahedron Lett. 1993, 34, 8267-8270.
T Minami; K Takahashi; T Hiyama; Tetrahedron Lett. 1993, 34, 3, 513-516.
DA Evans; AH Hoveyda; J. Org. Chem. 1990, 55, 5190-5192.
J Castorer; LA Sorbera; PA Leeson; Drugs Fut. 23(8), 1998, 847-859.
T Hiyama; K Takahashi; T Minami; Bull. Chem. Soc. Jpn. 1995, 68, 364-372.
MS Reddy; M Bairy; K Reddy; Oriental Journal of Chemistry. 2007, 23, 559-564.
RN Moore; G Bigam; JK Chan; AM Hogg; JC Vederas; J. Am. Chem. Soc. 1985, 107 3694-3701.
DS Johnson; JJ Li; Art of Drug Synthesis, John Wiley & Sons, New Jersey, 2007, 177-181.
MT Stone; Organic Lett. 2011, 13, 2326-2329.
SR Manne, SR Maramreddy, WO2007132482 (A2), 2007.
SD Dwivedi, DJ Patel, AP Shah, Cadila Healthcare Ltd, US0022102 (A1), 2012.
AM Gotta; LC Smith; IXth International Symp. Drugs Affecting Lipid Metabolism, 1986, 30- 31
Y Fujikawa, Nissan Chemical Industries Ltd, EP304063 (A3), 1989.
S Ahmed; CS Madsen; PD Stein; J. Med. Chem. 2008, 51, 2722-2733.
K Turner; Org. Process. Res. Dev, 2004, 8, 823-833.
Z Casar; M Steinbocher; J Kosmrlj; J. Org. Chem. 2010, 75(19), 6681-6684.
KL Baumann; Tetrahedron Letters, 1992, 33, 2283-2284.
N Miyachi; Y Yanagawa; H Iwasaki; Tetrahedron Lett. 1993, 34, 8267-8270.
T Minami; K Takahashi; T Hiyama; Tetrahedron Lett. 1993, 34, 3, 513-516.
DA Evans; AH Hoveyda; J. Org. Chem. 1990, 55, 5190-5192.
J Castorer; LA Sorbera; PA Leeson; Drugs Fut. 23(8), 1998, 847-859.
T Hiyama; K Takahashi; T Minami; Bull. Chem. Soc. Jpn. 1995, 68, 364-372.
MS Reddy; M Bairy; K Reddy; Oriental Journal of Chemistry. 2007, 23, 559-564.
RN Moore; G Bigam; JK Chan; AM Hogg; JC Vederas; J. Am. Chem. Soc. 1985, 107 3694-3701.
DS Johnson; JJ Li; Art of Drug Synthesis, John Wiley & Sons, New Jersey, 2007, 177-181.
MT Stone; Organic Lett. 2011, 13, 2326-2329.
SR Manne, SR Maramreddy, WO2007132482 (A2), 2007.
SD Dwivedi, DJ Patel, AP Shah, Cadila Healthcare Ltd, US0022102 (A1), 2012.
……………………………………………………..

……………………………

The reaction of 1 (R) ,7,7-trimethylbicyclo [2.2.1] heptan-2-one (I) with 1 -naphthylmagnesium bromide (II) gives the tertiary alcohol (III), which by reaction with SOCl2 and then with NaHCO3 yields 2 – (1-naphthyl) -1 (R) ,7,7-trimethylbicyclo [2.2.1] heptene (IV ). Hydroboration of (IV) with BH3 followed by oxidation with H2O2 affords 4 (S) ,7,7-trimethyl-3exo-(1-naphthyl) bicyclo [2.2.1] heptan-2exo-ol (V), which is submitted to transesterification with methyl acetoacetate (VI) and dimethyl-aminopyridine (DMAP) to give the corresponding ester (VII). The condensation of (VII) with N-methoxy-N-methyl-3-[2-cyclopropyl-4-( 4-fluorophenyl) quinolin-3-yl] -2 (E)-propenamide (VIII) by means of NaH yields the corresponding chiral 3,5-dioxoheptenoic acid ester (IX), which is selectively reduced first with diisobutylaluminum hy-dride acid (DIBAL) and then with diethylmethoxyborane and sodium borohydride affording the 3 (R), 5 (S)-dihydroxyheptenoic ester (X). Finally, this compound is saponified with NaOH and treated with acetic acid / sodium acetate. The intermediate amide (VIII ) is obtained by condensation of 2-cyclopropyl-4-(4-fluorophenyl) quinoline-3-carbaldehyde (XI) with N-methoxy-N-methylacetamide (XII) by means of butyllithium to the hydroxy propionamide (XIII), which is then dehydrated with methanesulfonyl chloride and triethylamine in the usual way).

…………………

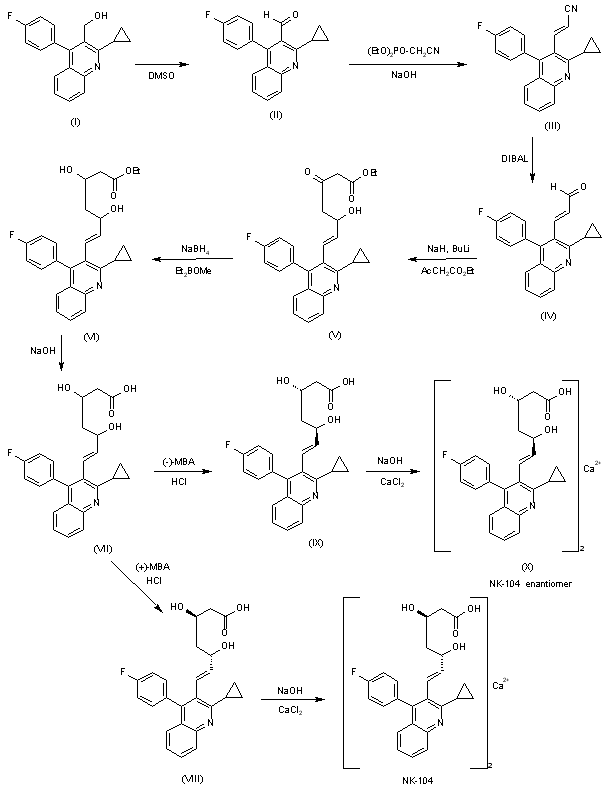
A systematic chiral synthesis of NK-104 and its enantiomer (X) has been reported: The oxidation of the already known 2-cyclopropyl-4-(4-fluorophenyl)quinoline-3-methanol (I) with DMSO, P2O5 and triethylamine gives the corresponding aldehyde (II), which is condensed with diethyl cyanomethylphosphonate by means of NaOH in toluene yielding the propenenitrile (III). The reduction of (III) with DIBAL affords the unsaturated aldehyde (IV), which is condensed with ethyl acetoacetate by means of NaH and n-BuLi to provide the 3-oxo-5-hydroxy-6-heptenoic acid ethyl ester derivative (V). The highly syn stereoselective reduction of (V) by means of diethylmethoxyborane and NaBH4 yields the desired syn racemic mixture of erythro-beta,delta-dihydroxyesters (VII), which is submitted to optical resolution with chiral (+)-alpha-methylbenzylamine [(+)-MBA] to obtain NK-104 free acid (VIII), which is finally treated with NaOH and CaCl2. The enantiomer of NK-104 has been obtained by optical resolution of the racemic mixture (VII) with (-)-alpha-methylbenzylamine to obtain the enantiomeric free acid (IX), which is treated with NaOH and CaCl2 as before.
Fujikawa, Y.; Suzuki, M.; Iwasaki, H.; Kitahara, M.; Sakashita, M.; Sakoda, R.;. Synthesis and biological evaluations of quinolone-based HMG-CoA reductase inhibitors Bioorg Med Chem 2001, 9 , 10, 2727

ADDITIONAL UPDATED INFO
Pitavastatin calcium is a novel member of the medication class of statins. Marketed in the United States under the trade name Livalo, it is like other statin drugs an inhibitor of HMG-CoA reductase, the enzyme that catalyses the first step of cholesterol synthesis. It is likely that pitavastatin will be approved for use in hypercholesterolaemia (elevated levels of cholesterol in the blood) and for the prevention of cardiovascular disease outside South and Southeast Asia as well.
Pitavastatin calcium is chemically known as (3R,5S)-7-[2-cyclopropyl-4-(4-fluorophenyl)quinolin-3-yl]-3,5-dihydroxy-6(E)-heptenoic acid calcium salt having the formula IA is known in the literature.
Pitavastatin is a synthetic lipid-lowering agent that acts as an inhibitor of 3-hydroxy-3-methylglutaryl-coenzyme a (HMG-CoA) reductase (HMG-CoA Reductase inhibitor). This enzyme catalyzes the conversions of HMG-CoA to mevalonate, inhibitors are commonly referred to as “statins”. Statins are therapeutically effective drugs used for reducing low density lipoprotein (LDL) particle concentration in the blood stream of patients at risk for cardiovascular disease. Pitavastatin is used in the treatment of hyperchloesterolemia and mixed dyslipidemia.
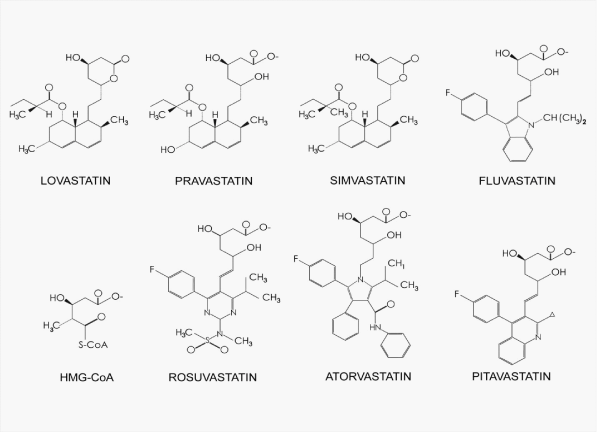
Pitavastatin calcium has recently been developed as a new chemically synthesized and powerful statin by Kowa Company Ltd, Japan. On the basis of reported data, the potency of Pitavastatin is dose-dependent and appears to be equivalent to that of Atorvastatin. This new statin is safe and well tolerated in the treatment of patients with hypercholesterolaemia. Significant interactions with a number of other commonly used drugs can be considered to be extremely low.
Pitavastatin was disclosed for the first time in US patents US 4,761,419, US 5,01 1 ,930 and US 5,753,675. The process disclosed in these patents for the preparation of Pitavastatin is as shown below:
wherein R is hydrogen or protecting group.
US 5,284,953 discloses a process for the preparation of Pitavastatin calcium, which employs optically active a-methylbenzylamine as a resoluting agent.
The above processes are economically not viable, as resolution is carried out in final stage.
US 6,835,838 B2 discloses a process for the preparation of Pitavastatin calcium, which is as shown below:
However, it has been observed that the above process of lactonization results in ~10- 15% of unreacted Pitavastatin ethyl ester and therefore results in low yield. Further, -10% of Pitavastatin acid results during the above lactonization process and therefore does not produce a single product which is required to keep adequate control for an intermediate through specifications to have consistently better quality of the finished product.
Processes for the preparation of Pitavastatin are described in EP-A-0304063 and EP-A-1099694 and in the publications by N. Miyachi et al. in Tetrahedron Letters (1993) vol. 34, pages 8267-8270 and by K. Takahashi et al. in Bull. Chem. Soc. Japan (1995) Vol. 68, 2649-2656. These publications describe the synthesis of Pitavastatin in great detail but do not describe the hemi-calcium salt of Pitavastatin. The publications by L A. Sorbera et al. in Drugs of the Future (1998) vol. 23, pages 847-859 and by M. Suzuki at al. in Bioorganic & Medicinal Chemistry Letters (1999) vol. 9, pages 2977-2982 describe Pitavastatin calcium, however, a precise procedure for its preparation is not given. A full synthetic procedure for the preparation of Pitavastatin calcium is described in EP-A-0520406. In the process described in this patent Pitavastatin calcium is obtained by precipitation from an aqueous solution as a white crystalline material with a melting point of 190-192° C.
US20090182008 A1 discloses polymorphic form A, B, C, D, E, and F, and the amorphous form of Pitavastatin Calcium salt (2:1). In particular, crystalline Form A having water content from about. 5% to about 15% and process for its preparation are disclosed.
US20090176987 A1 also discloses polymorphic form crystal form A of Pitavastatin Calcium which contains from 5 to 15% of water and which shows, in its X-ray powder diffraction as measured by using CuKa radiation, a peak having a relative intensity of more than 25% at a diffraction angle (20) of 30.16°.
WO2007/132482 A1 discloses a novel process for the preparation of Pitavastatin Calcium by condensing bromide salt of formula-3 with aldehyde compound of formula-4 to obtain olefinic compound of formula-5 and converting olefinic compound to Pitavastatin Calcium via organic amine salt for purification.
Pitavastatin and its process were disclosed in U.S. Pat. No. 5,753,675.
Pitavastatin calcium and its process were disclosed in U.S. Pat. No. 5,856,336. PCT publication no. WO 2004/072040 (herein after referred to ’040 patent) disclosed crystalline polymorph A, polymorph B, polymorph C, polymorph D, polymorph E, polymorph F and amorphous form of pitavastatin calcium
- Synthesis of pitavastatin via cross-coupling reaction is disclosed inTetrahedron Lett. 1993, 34, 8263-8266, and in Tetrahedron Lett. 1993, 34, 8267-8270.
- A method for the preparation of pitavastatin via epichlorohydrin is described in Tetrahedron: Asymmetry 1993, 4, 201-204.
- PCT application WO 2003/064382 describes a method for preparation of pitavastatin by asymmetric aldol reaction, in which titanium complex is used as a catalyst.
- HWE route to pitavastatin by utilization of 3-formyl substituted pitavastatin heterocycle is disclosed in Helv. Chim. Acta 2007, 90, 1069-1081:
- Methods for preparation of pitavastatin heterocycle derivatives are described in Bull. Chem. Soc. Jpn. 1995, 68, 364-372, Heterocycles 1999, 50, 479-483, Lett. Org. Chem. 2006, 3, 289-291, and in Org. Biomol. Chem. 2006, 4, 104-110, as well as in the international patent applications WO 95/11898 and WO 2004/041787
WO 1995/1 1898 Al discloses a process for the preparation of Pitavastatin, which is as shown below:
wherein Y represents P+RnRi2Ri3Hal“ or P(W)Ri4R15; R9a, R% and R]0 are protecting groups each of Rn, Rj2> R^, Ri4 and R15 which are independent of one another, is optionally substituted alkyl or optionally substituted aryl group; R14 and Rj5 together form a 5- or 6-membered ring; Hal is chlorine, bromine or iodine; and W is O or S.
The above process results in 2-5% of Cis isomer of Pitavastatin which requires further purification and therefore results poor yield.
US 6,875,867 B2 discloses a process for the preparation of Pitavastatin arginine salt, which is as shown below:
Saponification / Base
During the above process Trifluoroacetic acid or hydrochloric acid is used to break the acetonide and the Pitavastatin ester formed is converted in situ to its corresponding alkali salt by treating with base, such as sodium hydroxide.
US20090182008 A1 discloses polymorphic form A, B, C, D, E, and F, and the amorphous form of Pitavastatin Calcium salt (2:1). In particular, crystalline Form A having water content from about. 5% to about 15% and process for its preparation are disclosed.
……………………………………..
nmr
calcium bis-(E)-3,5-dihydroxy-7-[4’-(4’’-flurophenyl)-2’-
cyclopropyl-quinoline-3-yl]-hept-6-enoate , pitavastatin calcium
Melting Point: 207 degC;
cyclopropyl-quinoline-3-yl]-hept-6-enoate , pitavastatin calcium
Melting Point: 207 degC;
IR υmax (KBr) cm-1: 3366 (OH), 2911, 1603 (C=O), 1567 (C=N), 1513 (C=C),
1488 (C-H), 1416 (C-H), 1313, 1275, 1221 (C-O-C), 1158, 1065 (C-H), 972, 843, 763.
1H-NMR (500MHz, DMSO-d6):
δ 1.01 (m, 2H), 1.09 (m, 1H), 1.19 (m, 2H), 1.41 (m, 1H),
1.98 (dd, 1H, J1 =8.5,
J2 =15.5Hz), 2.11(d, 1H, J1 =3.0, J2 =15.5Hz), 2.50 (m, 2H),
J2 =15.5Hz), 2.11(d, 1H, J1 =3.0, J2 =15.5Hz), 2.50 (m, 2H),
3.66 (m, 1H), 4.13 (m, 1H), 4.95 (s, 1H), 5.58 (dd, 1H,
J1 =5.5, J2 =10.5Hz), 6.49 (d, 1H, J = 16.0Hz),
J1 =5.5, J2 =10.5Hz), 6.49 (d, 1H, J = 16.0Hz),
7.35 (m, 6H), 7.59 (m, 1H, J = 7.0Hz), 7.83 (d, 1H, J =8.5Hz).
13CNMR & DEPT (125.76MHz, DMSO-d6):
δ 11.12(CH2, C-17), 11.23(CH2,C-18), 15.80(CH2, C-16), 44.29(CH2,
C-22), 44.61(CH2, C-24), 66.61(C-O, C-23),
C-22), 44.61(CH2, C-24), 66.61(C-O, C-23),
69.34(C-O,C-21),115.53(C=C, C-20), 15.62(CH), 115.79(CH),
123.59(CH), 126.07(C=C, C-19),
123.59(CH), 126.07(C=C, C-19),
128.79(CH),129.20(CH),130.07(CH), 32.30(CH),
132.56(CH), 133.51(C),
142.60(C), 144.09(C), 146.37(C),
142.60(C), 144.09(C), 146.37(C),
161.02(C), 163.00(C), 179.13(C=O, C-25).
ESI-MS: m/z (%) 318 (100), 274 (23), 423 (13), 422 (M+, 70); EI calcd for C25H24FNO4, 421.461; found, 422.220
(M+).
(M+).
…………………


......................................
2 ATORVASTATIN

atorvastatin

Atorvastatin (INN) /əˌtɔrvəˈstætən/, marketed by Pfizer as a calcium salt under the trade name Lipitor,[1] is a member of the drug class known as statins, used for lowering blood cholesterol. It also stabilizes plaque and prevents strokes through anti-inflammatory and other mechanisms. Like all statins, atorvastatin works by inhibitingHMG-CoA reductase, an enzyme found in liver tissue that plays a key role in production of cholesterol in the body.

Atorvastatin was first synthesized in 1985 by Bruce Roth of Parke-Davis Warner-Lambert Company (since acquired by Pfizer). The best selling drug in pharmaceutical history, sales of Lipitor since it was approved in 1996 exceed US$125 billion, and the drug has topped the list of best-selling branded pharmaceuticals in the world for nearly a decade.[2] When Pfizer's patent on Lipitor expired on November 30, 2011,[3] generic atorvastatin became available in the United States, initially manufactured only by generic drugmakers Watson Pharmaceuticals and India's Ranbaxy Laboratories. Prices for the generic version did not drop to the level of other generics—$10 or less for a month's supply—until other manufacturers were able to supply the drug in May 2012.[4]

| Atorvastatin calcium, YM-548, CI-981, Prevencor, Tahor, Lipibec, Torvast, Sortis, Lipitor | |
| (3R,5R)-7-[2-(4-Fluorophenyl)-5-isopropyl-3-phenyl-4-(phenylcarbamoyl)pyrrol-1-yl]-3,5-dihydroxyheptanoic acid calcium salt (2:1) | |
| 134523-03-8, 134523-00-5 (free acid), 110862-48-1 (free acid (R*,R*)-isomer) | |
| 2-C33-H34-F-N2-O5.Ca | |
| 1155.35 | |
| Alzheimer's Dementia, Treatment of , Cardiovascular Drugs, Cognition Disorders, Treatment of, Immunologic Neuromuscular Disorders, Treatment of, Lipoprotein Disorders, Treatment of , Metabolic Drugs, Multiple Sclerosis, Agents for, Neurologic Drugs, Treatment of Disorders of the Coronary Arteries and Atherosclerosis, HMG-CoA Reductase Inhibitors, TNFSF6 Expression Inhibitors | |
| Launched-1997 | |
| Jouveinal (Originator), Pfizer (Originator), Almirall Prodesfarma (Licensee), Syncro (Licensee), Yamanouchi (Licensee), Stanford University (Codevelopment) |
| SYNTHESISTrans-6-[2-(3- or 4-carboxamido-substd. pyrrol-1-yl)alkyl]-4-hydroxypyran-2-one inhibitors of cholesterol synthesis | |
| Roth, B.D. (Pfizer Inc.) | |
| EP 0247633; US 4681893 | |
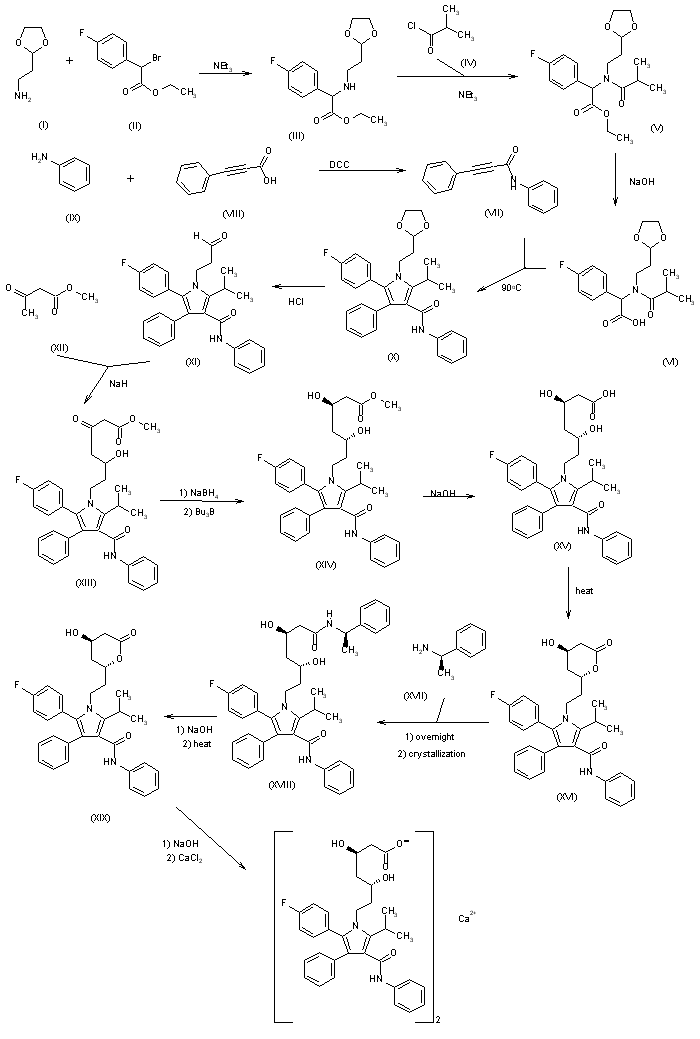 | |
1) The condensation of 2-(1,3-dixolan-2-yl)ethylamine (I) with ethyl 2-bromo-2-(4-fluorophenyl)acetate (II) by means of triethylamine in acetonitrile gives ethyl 2-[2-(1,3-dioxolan-2-yl)ethylamino]-2-(4-fluorophenyl)acetate (III), which is acylated with isobutyryl chloride (IV) and triethylamine in dichloromethane yielding the corresponding amide (V). Saponification of the ester (V) with NaOH in methanol/water affords the free acid (VI), which is cyclized with N,3-diphenylpropynamide (VII) [obtained in the reaction of 3-phenylpropynoic acid (VIII) with aniline (IX) by means of dicyclohexylcarbodiimide (DCC)] by heating at 90 C in acetic anhydride giving 1-[2-(1,3-dioxolan-2-yl)ethyl]-5-(4-fluorophenyl)-2-isopropyl-N,4-diphenylpyrrole-3-carboxamide (X). The hydrolysis of the dioxolane group of (X) with HCl yields the corresponding aldehyde (XI), which is condensed with methyl acetoacetate (XII) by means of NaH in THF affording 7-[2-(4-fluorophenyl)-5-isopropyl-3-phenyl-4-(N-phenylcarbamoyl)pyrrol-1-yl]-5-hydroxy-3-oxoheptanoic acid methyl ester (XIII). The reduction of the carbonyl group of (XIII) with tributylborane and NaBH4 in THF gives the (3R*,5R*)-dihydroxy ester (XIV), which is saponified with NaOH in water yielding the corresponding free acid (XV). The lactonization of (XV) by heating in refluxing toluene affords the (R*,R*)-lactone (XVI), which is submitted to optical resolution by reaction with (R)-1-phenylethylamine (XVII) followed by fractional crystallization thus obtaining the amide (XVII) as the pure (R,R,R)-enantiomer. The hydrolysis of the amide (XVIII) with NaOH, followed by heating in refluxing toluene gives the (R,R)-lactone (XIX), which is finally treated first with NaOH in methanol/water, and then with CaCl2 or calcium acetate. | |
- "Pfizer product promotion page (Liptor)". Retrieved 2011-12-05.
- "Lipitor becomes world's top-selling drug". Crain's New York Business. 2011-12-28.
- CNN Wire Staff (November 30, 2011). "Lipitor loses patent, goes generic". CNN. Retrieved November 18, 2012.
- NeLM, June 2012: Price to UK National Health Service for 28 tablets from £3.25 (10mg) to £10.00 (80mg).
- "Atorvastatin Calcium". Drugs.com. Retrieved 3 April 2011.

Further reading
- "Highlights of prescribing information" (pdf). Lipitor (atorvastatin calcium) Tablets for oral administration. Pfizer. 2009-06-01. Retrieved 2011-10-26.
- Maggon K (June 2005). "Best-selling human medicines 2002-2004". Drug Discov. Today 10 (11): 739–42. doi:10.1016/S1359-6446(05)03468-9. PMID 15922927.
- Roth BD (2002). "The discovery and development of atorvastatin, a potent novel hypolipidemic agent". Prog Med Chem. Progress in Medicinal Chemistry 40: 1–22. doi:10.1016/S0079-6468(08)70080-8. ISBN 978-0-444-51054-9. PMID 12516521.
- Simons J (2003-01-20). "The $10 Billion Pill Hold the fries, please. Lipitor, the cholesterol-lowering drug, has become the bestselling pharmaceutical in history. Here's how Pfizer did it". Fortune. Retrieved 2011-10-26.
- Winslow R (2000-01-24). "The Birth of a Blockbuster: Lipitor's Route out of the Lab". The Wall Street Journal. Retrieved 2011-10-26.
- "Ann Arbor chemist wins national award for drug discovery". ScienceBlog. American Chemical Society. 2003-03-01. Retrieved 2011-10-26.
- Rowe A (2008-08-20). "Meet the Guy Who Invented Lipitor". Wired Science. Wired.com. Retrieved 2011-10-26.
- Bernstein M (2008-08-16). "Chemical Society To Honor 'Heroes Of Chemistry' During National Meeting". Medical News Today. Retrieved 2011-10-26.
- He L (2003-09-27). "Bruce D. Roth, Pfizer Inc, USA". Chinese Academy of Sciences·Institute of Process Engineering. Retrieved 2011-10-26.
External links
- Atorvastatin bound to proteins in the PDB
- Lipitor.com – manufacturer's site
- MedlinePlus Drug information: Atorvastatin (Systemic) – information from USP DI Advice for the Patient
- U.S. National Library of Medicine: Drug Information Portal - Atorvastatin
| An improved synthesis of 1,1-dimethylethyl 6-cyanomethyl-2,2-dimethyl-1,3-dioxane-4-acetate, a key intermediate for atorvastatin synthesis | |
| Radl, S.; et al. | |
| Tetrahedron Lett 2002,43(11),2087 | |
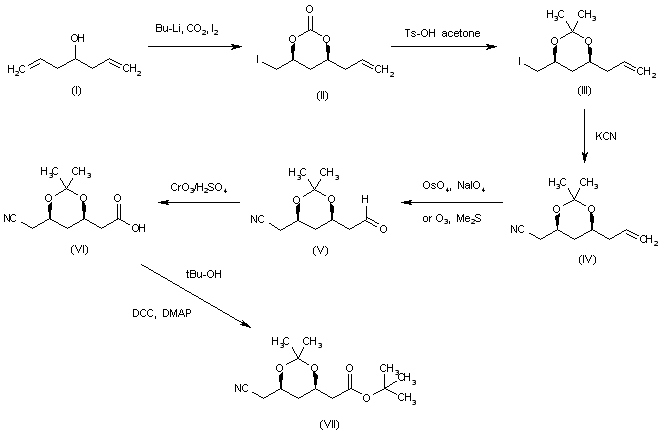 | |
The 6-cyanohexanoic ester (VII), intermediate in the synthesis of 180072 (see intermediate (XLI) in scheme no. 18007204a) has been obtained as follows: the reaction of 1,6-heptadien-4-ol (I) with BuLi, CO2, and I2 in THF gives the cyclic carbonate (II), which is treated with Ts-OH in acetone to yield the acetonide (III). The reaction of the iodine atom of (III) with KCN in hot DMSO affords the nitrile (IV), which is oxidized at the terminal double bond with OsO4 and NaIO4, or O3 and Me2S, to provide the carbaldehyde (V). The oxidation of (V) with CrO3/H2SO4 in acetone gives the carboxylic acid (VI), which is finally esterified with tert-butanol by means of DCC and DMAP in dichloromethane, yielding the target ester intermediate (VII). | |
Lipitor industrial production shown below (TL, 1985, 2951; TL, 1992, 2279; 2283)
Divided into two fragments. Primary amine fragment iso-ascorbic acid as a starting material (stereoisomer of vitamin C) in the system. 1,4 - dione as a starting material isobutyrylacetanilide fragment, obtained by the Stetter reaction, the reaction of benzoin conjugated version Michael addition. Diketone related primary amine with a substituted pyrrole ring obtained five, after deprotection and salt formation Lipitor.
Atorvastatin of IC50-0.025μM, its RR configuration is as high IC50-0.007μM (SS configuration IC50-0.44μM).
Initially synthesized as shown above (JMC, 1991, 357).
Which polysubstituted pyrrole ring by the Munchnone of 1,3-dipolar [3 +2] cycloaddition get.
Which polysubstituted pyrrole ring by the Munchnone of 1,3-dipolar [3 +2] cycloaddition get.
(atorvastatin), a part of statin (statins) a class of drugs.

.....................
3.
..........................................

............................
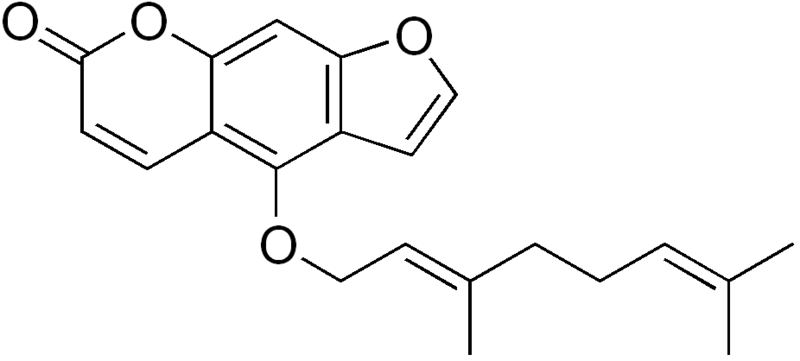
CITRUS AND STATINS
bergamottin
Citrus And Statins. —Citrus And Statins “” “How Foods and Drugs Collide” provided my first knowledge that 5-geranoxy psoralen (bergamottin) had been identified as the active agent in grapefruit juice implicated in interfering with the so-called statin drugs
( C&ENCitrus And Statins, Sept. 27, page 55 William L. Stanley Carmel, Calif. /articles/88/i49/Citrus-Statins.html 20101206 88 49 /magazine/88/8849.html /departments/letters.html Citrus And Statins Letters Citrus And Statins Chemical & Engineering News Citrus And Statins ACS 2009 IRS Form 990 Available The American Chemical Society’s 2009 Form 990 is now available on ACS’s website.
Chemical & Engineering News, 88(49), December 06, 2010 [Letters] you will have to pay a fee or subscribe
free info from net
Bergamottin is a natural furanocoumarin found principally in grapefruit juice. It is also found in the oil of bergamot orange, from which it was first isolated and from which its name is derived. To a lesser extent, bergamottin is also present in the essential oils of other citrus fruits. Along with the chemically related compound 6′,7′-dihydroxybergamottin, it is believed to be responsible for thegrapefruit juice effect in which the consumption of the juice affects the metabolism of a variety of pharmaceutical drugs.[1]
Chemistry
Chemically, bergamottin and dihydroxybergamottin are linear furanocoumarins functionalized with side chains derived fromgeraniol. They are inhibitors of some isoforms of the cytochrome P450 enzyme, particularly CYP3A4.[2] This prevents oxidative metabolism of certain drugs by the enzyme, resulting in an elevated concentration of drug in the bloodstream.
Normally, the grapefruit juice effect is considered to be a negative interaction, and patients are often warned not to consume grapefruit or its juice when taking medication. However, some current research is focused on the potential benefits of cytochrome P450 inhibition.[3] Bergamottin, dihydroxybergamottin, or synthetic analogs may be developed as drugs that are targeted to increase the oral bioavailability of other drugs. Drugs that may have limited use because they are metabolized by CYP3A4 may become viable medications when taken with a CYP3A4 inhibitor because the dose required to achieve a necessary concentration in the blood would be lowered.[4]
Biosynthesis of bergamottin
Bergamottin is derived from components originating in the shikimate pathway.[5] The biosynthesis of this compound starts with the formation of the demethylsuberosin (3) product which is formed via the alkylation of the umbelliferone (2) compound.[6] The alkylation of the umbelliferone is initiated with the use of dimethylallyl pyrophosphate, more commonly known as DMAPP. The cyclization of an alkyl group occurs to form marmesin (4), which is done in the presence of NADPH and oxygen along with a cytochrome P450 monooxygenase catalyst.[7] This process is then repeated twice more, first to remove the hydroxyisopropyl substituent from marmesin (4) to form psoralen (5), and then to add a hydroxyl group to form bergaptol (6).[8] Bergaptol (6) is next methylated with SAM, S-Adenosyl methionine, to form bergapten (7). The final step in this biosynthesis is the attachment of a GPP, or geranyl pyrophosphate, to the newly methylated bergapten (7) to form the target molecule bergamottin (8).
References
- ^ David G. Bailey, J. Malcolm, O. Arnold, J. David Spence (1998). “Grapefruit juice-drug interactions”. Br J Clin Pharmacol 46 (2): 101–110. doi:10.1046/j.1365-2125.1998.00764.x.PMC 1873672. PMID 9723817.
- ^ Basavaraj Girennavar, Shibu M. Poulose, Guddadarangavvanahally K. Jayaprakasha, Narayan G. Bhat and Bhimanagouda S. Patila (2006). “Furocoumarins from grapefruit juice and their effect on human CYP 3A4 and CYP 1B1 isoenzymes”. Bioorganic & Medicinal Chemistry 14 (8): 2606–2612. doi:10.1016/j.bmc.2005.11.039. PMID 16338240.
- ^ E. C. Row, S. A. Brown, A. V. Stachulski and M. S. Lennard (2006). “Design, synthesis and evaluation of furanocoumarin monomers as inhibitors of CYP3A4″. Org. Biomol. Chem. 4(8): 1604–1610. doi:10.1039/b601096b. PMID 16604230.
- ^ Christensen, Hege; Asberg, Anders; Holmboe, Aase-Britt; Berg, Knut Joachim (2002). “Coadministration of grapefruit juice increases systemic exposure of diltiazem in healthy volunteers”. European Journal of Clinical Pharmacology 58 (8): 515–520. doi:10.1007/s00228-002-0516-8. PMID 12451428.
- ^ Dewick, P. Medicinal Natural Products:A Biosynthetic Approach, 2nd ed., Wiley&Sons: West Sussex, England, 2001, p 145.
- ^ Bisagni, E. Synthesis of psoralens and analogues. J. Photochem. Photobiol. B. 1992, 14, 23-46.
- ^ Voznesensky, A. I.; Schenkman, J. B. The cytochrome P450 2B4-NADPH cytochrome P450 reductase electron transfer complex is not formed by charge-pairing. J. Biol. Chem. 1992, 267, 14669-14676.
- ^ Kent, U. M.; Lin, H. L.; Noon, K. R.; Harris, D. L.; Hollenberg, P. F. Metabolism of bergamottin by cytochromes P450 2B6 and 3A5. J. Pharmacol. Exp. Ther. 2006, 318, 992-1005














Over eight years of successful operation in the pharmaceutical industry has enabled us to possess a full-bodied array of building blocks in numerous pack sizes. Pitavastatin Calcium
ReplyDelete