1 IDRAPARINUX
2 FONDAPARINUX
3
4

1 IDRAPARINUX

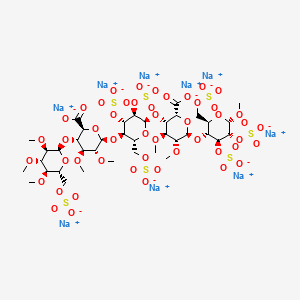 IDRAPARINUX
IDRAPARINUX




























2 FONDAPARINUX
3
4

1 IDRAPARINUX

IDRAPARINUX
Nonasodium (2S,3S,4S,5R,6R)-6-[(2R,3R,4S,5R,6R)-6-[(2R,3S,4S,5R,6R)-2-carboxy-4,5-dimethoxy-6-[(2R,3R,4S,5R,6S)-6-methoxy-4,5-disulfooxy-2-(sulfooxymethyl)oxan-3-yl]oxyoxan-3-yl]oxy-4,5-disulfooxy-2-(sulfooxymethyl)oxan-3-yl]oxy-4,5-dimethoxy-3-[(2R,3R,4S,5R,6R)-3,4,5-trimethoxy-6-(sulfooxymethyl)oxan-2-yl]oxyoxane-2-carboxylic acid |
| CAS NUMBER | 149920-56-9 |
|---|
| FORMULA | C38H55Na9O49S7 |
|---|---|
| MOL. MASS | 1727.17683 g/mol |
CAS 162610-17-5 (free acid)
SANORG34006, SR-34006, SanOrg 34006, SanOrg-34006, UNII-H84IXP29FN, AC1MJ0N4, Org-34006
Methyl O-2,3,4-tri-O-methyl-6-O-sulfo-alpha-D-glucopyranosyl-(1–4)-O-2,3-di-O-methyl-beta-D-glucopyranuronosyl-(1–4)-O-2,3,6-tri-O-sulfo-alpha-D-glucopyranosyl-(1–4)-O-2,3-di-O-methyl-alpha-L-idopyranuronosyl-(1–4)-2,3,6-tri-O-sulfo-alpha-D-glucopyran
Methyl O-2,3,4-tri-O-methyl-α-D-glucopyranosyl-(1→4)-O-2,3-di-O-methyl-β-D-glucopyranosyluronic acid-(1→4)-O-α-D-glucopyranosyl-(1→4)-O-2,3-di-O-methyl-α-L-idopyranosyluronic acid-(1→4)-O-α-D-glucopyranose
methyl O-2,3,4-tri-O-methyl-6-O-sulfo-α-D-glucopyranosyl-(1→4)-O-2,3-di-O-methyl-α-L-idopyranuronosyl-(1→4)-O-2,3,6-tri-O-sulfo-α-D-glucopyranosyl-(1→4)-O-2,3-O-di-methyl-α-L-idopyranuronosyl-(1→4)-O-2,3,6-tri-O-sulfo-α-D-glucopyranoside nonakis sodium salt. [α]D²⁰ = +46.2° (c=1; water). Anomeric protons chemical shifts: 5.43; 5.37; 5.16; 5.09; and 5.09 ppm.
Idraparinux sodium, or methyl O-2,3,4-tri-O-methyl-6-O-sodium sulfonato-α-D-glucopyranosyl-(1→4)-O-2,3-di-O-methyl-β-D-glucopyranosyluronate sodium-(1→4)-O-2,3,6-tri-O-sodium sulfonato-α-D-glucopyranosyl-(1→4)-O-2,3-di-O-methyl-α-L-idopyranosyluronate sodium-(1→4)-O-2,3,6-tri-O-sodium sulfonato-α-D-glucopyranose, is a pentasaccharide with antithrombotic activity.
The preparation of idraparinux by sulfatation of a deprotected pentasaccharide is described in Bioorganic & Medicinal Chemistry, 1994, Vol. 2, No. 11, pp. 1267-1280, and also in patent EP 0 529 715 B1.
It has a similar chemical structure and the same method of action as fondaparinux, but with an elimination half-life about five to six times longer (an increase from fondaparinux’s 17 hours to approximately 80 hours), which means that the drug should only need to be injected once a week.
As of July 2007, it has completed the Phase III clinical trial AMADEUS.
Idraparinux selectively blocks coagulation factor Xa.[2]
See Heparin: Mechanism of anticoagulant action for a comparison of the mechanism of heparin, low-molecular-weight heparins, fondaparinux and idraparinux.
Idraparinux sodium is a synthetic pentasaccharide with indirect coagulation factor Xa inhibitor activity. The drug candidate had been in phase III clinical development at Sanofi (formerly known as sanofi-aventis) for the once-weekly long-term treatment and secondary prevention of venous thromboembolic events in patients with pulmonary embolism (PE) and deep vein thrombosis (DVT), as well as for the prevention of thromboembolic complications related to atrial fibrillation (AF).
However, no recent development has been reported for this research. The oligosaccharide is delivered by subcutaneous injection. Unlike other products, idraparinux is administered once weekly rather than daily, thereby increasing patient convenience.
Originally developed under a collaboration between sanofi-sventis and Akzo Nobel’s human healthcare business Organon, all rights to idraparinux were transferred to Sanofi in January 2004 in exchange for revenues based on future sales.
 IDRAPARINUX
IDRAPARINUX
Several synthetic pentasaccharides have been developed, such as Idraparinux, where all hydroxyl groups are methylated or sulphated, as illustrated below:
Initially, the firm Organon developed a way of synthesis for the preparation of the “active pentasaccharide”. This synthesis, using the 3-0-benzyl-1 ,2-0-isopropylidene-a-D- glucofuranose as substrate (Van Boeckel et al., J. Carbohydr. Chem. 1985, 4, p.293-321 ), comprises more than 50 steps, and the inversion of configuration of the C5 carbon is carried out by the opening of an epoxide. After a step of protection followed by a bromination, the G unit is thus obtained. It is well known that the synthesis of said G unit is very tedious, due to the number of steps for obtaining such unit and the known tendency of L-idose derivatives to exist as furanoses. After being coupled to the H unit, successive steps of protection-deprotection then an oxidation reaction carried out on C6 carbon, lead to the GH disaccharide.
In the preparation of Idraparinux, the synthesis of the disaccharide GH is nearly similar to the above synthesis of early synthetic pentasaccharides. The major innovation lies in the obtaining of disaccharide EF by epimerization of disaccharide GH. The coupling of both disaccharides leads to the tetrasaccharide EFGH, which is further coupled to the D unit for obtaining said pentasaccharide. The preparation of the disaccharide EF from GH allows notably the decrease of the total number of the steps to approximatively 25 (Petitou, M.; Van Boeckel, C.A. Angew. Chem., Int.Ed. 2004, 43, p.31 18-3133).
Hence, all current syntheses of the “active pentasaccharide” comprise a large number of steps and more particularly involves the complex synthesis of key L-iduronic acid derivative (G unit). Indeed, the preparation of the G unit of the “active pentasaccharide” of heparin has always been a limiting step in the synthesis of antithrombotic heparin derivatives.
Thus, there is still a need for a new efficient process of preparation of L-iduronic acid derivative, which would not possess the drawbacks established above and would be compatible with industrial scales. Besides, there is a need for such process which would in addition lead to an improved process of preparation of the “active pentasaccharide” constituting the heparin derivatives.
- Idrabiotaparinux, developed by sanofi-aventis, is the biotinylated pentasaccharide corresponding to the structure depicted below. The pentasaccharide structure of idrabiotaparinux is the same as idraparinux, another antithrombotic agent developed by sanofi-aventis (see structure below). However in idrabiotaparinux, the presence of a biotin hook covalently linked to the first saccharidic unit enables the compound to be neutralized by avidin or streptavidin, as described in the international patent application WO 02/24754 .
- In the EQUINOX trial, which enrolled patients with DVT treated for 6 months with equimolar doses of either idrabiotaparinux or idraparinux, idrabiotaparinux, with the same anti-activated factor X pharmacological activity (hereafter “anti-Xa activity”) as idraparinux, was shown to have a similar efficacy, but, surprisingly, a better safety with less observed bleedings, in particular major bleedings.
- Therefore, the subject-matter of the invention is the use of idrabiotaparinux for the manufacture of a medicament useful for the treatment and secondary prevention of thrombotic pathologies, wherein the use of idrabiotaparinux involves a decrease in the incidence of bleedings during said treatment.
- In other words, the invention relates to the use of idrabiotaparinux as an antithrombotic treatment, wherein said use minimizes the risk of bleedings during the antithrombotic treatment. Indeed, idrabiotaparinux enables to increase the benefit-risk ratio during the antithrombotic treatment.
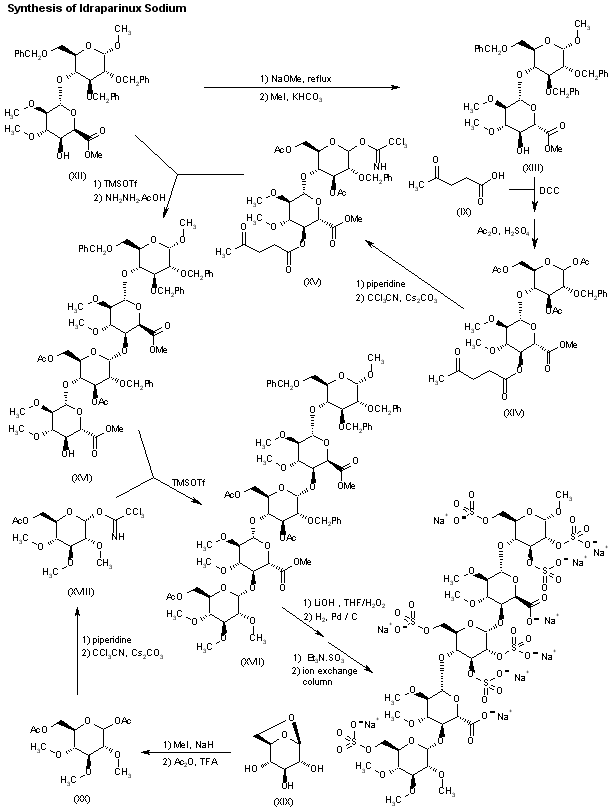
The L-ioduronic acid methyl ester derivative (XII) is then converted into its D-glucuronic acid methyl ester counterpart (XIII) by epimerization with NaOMe in refluxing MeOH, followed by esterification with MeI and KHCO3 in DMF.
Protection of the ester (XIII) with levulinic acid (IX) by means of DCC and DMAP in dioxane, followed by acetolysis of the anomeric center with sulfuric acid in acetic anhydride furnishes the disaccharide (XIV), which is then saponified with piperidine and subjected to reaction with trichloroacetonitrile and Cs2CO3 in THF to yield the imidate (XV).
Glycosylation of the disaccharide (XII) with the imidate (XV) by means of trimethylsilyl triflate in CH2Cl2, followed by removal of the levulinoyl group by means of hydrazine acetate, furnishes the tetrasaccharide (XVI), which is coupled with the glucosyl trichloroacetimidate (XVIII) by means of trimethylsilyl trifluoromethanesulfonate in CH2Cl2 providing the pentasaccharide (XVII).
Glucosyl imidate (XVIII) is prepared by methylation of 1,6-anhydroglucose (XIX) with MeI and NaH in DMF, followed by acetolysis with Ac2O/TFA to give compound (XX), which is treated with piperidine in THF and finally with trichloroacetonitrile in dichloromethane in the presence of Cs2CO3.
The pentasaccharide (XVII) is deprotected by saponification with LiOH in THF/H2O2, and then hydrogenated over Pd/C in tert-butanol/water to provide a fully deprotected pentamer, which is finally subjected to sulfation with triethylamine sulfur trioxide complex in DMF and converted into the corresponding sodium salt by elution in a Dowex 50 XW4-Na+ or a Mono-Q anion-exchange column.


……………..
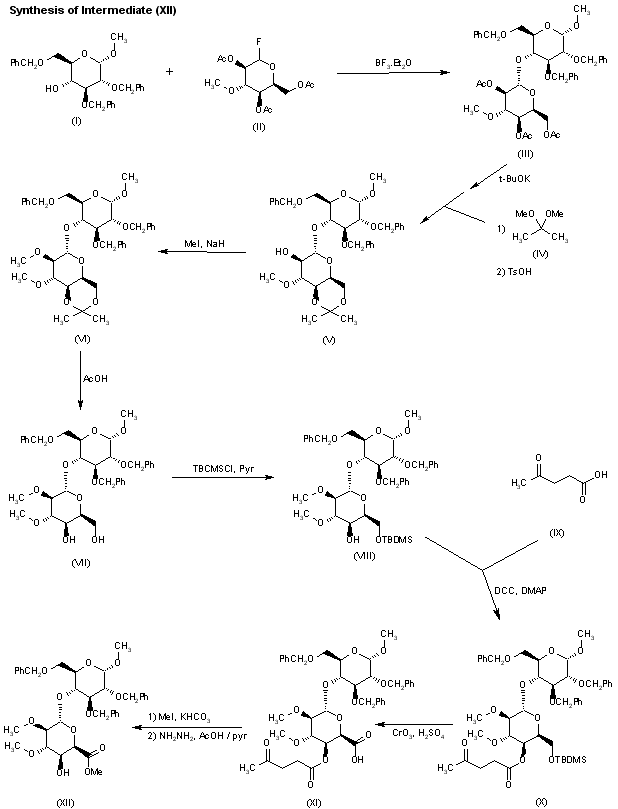
Glycosylation of sugar (I) with the idopyranosyl fluoride (II) by means of BF3.Et2O and molecular sieves in dichloromethane gives the disaccharide fragment (III), which is then converted into acetonide (V) by saponification of the ester functions with t-BuOK, followed by reaction with 2,2-dimethoxypropane (IV) in DMF and acidification with p-toluensulfonic acid. Methylation of acetonide (V) with MeI and NaH in DMF/MeOH provides the disaccharide (VI), which is then treated with HOAc to yield the 4′,6′-diol (VII). Selective silylation of the diol (VII) with tert-butyldimethylsilyl chloride (TBDMSCl) in pyridine leads to the 6′-O-TBDMS derivative (VIII), which is condensed with levulinic acid (IX) by means of dicyclohexylcarbodiimide (DCC) and 4-dimethylaminopyridine (DMAP) in dioxane to give the ester (X). Compound (X) is then submitted to simultaneous Jones oxidation and TBDMS removal with CrO3 and H2SO4/H2O in acetone to provide the iduronic acid derivative (XI), which is converted into the key intermediate (XII), first by esterification with MeI and KHCO3 in DMF and then by removal of the 4′-O-levulinoyl protecting group with HOAc and hydrazine hydrate in pyridine.


………………………
Idraparinux sodium, or methyl O-2,3,4-tri-O-methyl-6-O-sodium sulfonato-α-D-glucopyranosyl-(1→4)-O-2,3-di-O-methyl-β-D-glucopyranosyluronate sodium-(1→4)-O-2,3,6-tri-O-sodium sulfonato-α-D-glucopyranosyl-(1→4)-O-2,3-di-O-methyl-α-L-idopyranosyluronate sodium-(1→4)-O-2,3,6-tri-O-sodium sulfonato-α-D-glucopyranose, is a pentasaccharide with antithrombotic activity.
The preparation of idraparinux by sulfatation of a deprotected pentasaccharide is described in Bioorganic & Medicinal Chemistry, 1994, Vol. 2, No. 11, pp. 1267-1280, and also in patent EP 0 529 715 B1.
A crystalline form of the pentasaccharide methyl O-2,3,4-tri-O-methyl-α-D-glucopyranosyl-(1→4)-O-2,3-di-O-methyl-β-D-glucopyranosyluronic acid-(1→4)-O-α-D-glucopyranosyl-(1→4)-O-2,3-di-O-methyl-α-L-idopyranosyluronic acid-(1→4)-O-α-D-glucopyranose has now been isolated. This compound in its crystalline form has proven to be very useful for the preparation of idraparinux, since it makes it possible to obtain this product in a particularly interesting chemical yield and with a significant gain in quality, the purity being improved as regards the crude product obtained, as will be detailed hereinbelow. These gains in reaction yield and in purity for the production of idraparinux are considerable advantages from an industrial viewpoint, since improving the robustness of a process is a constant cause for concern, especially in the case of large-scale syntheses.
One subject of the invention is thus the compound methyl O-2,3,4-tri-O-methyl-α-D-glucopyranosyl-(1→4)-O-2,3-di-O-methyl-β-D-glucopyranosyluronic acid-(1→4)-O-α-D-glucopyranosyl-(1→4)-O-2,3-di-O-methyl-α-L-idopyranosyluronic glucopyranose in crystalline form.
Methyl O-2,3,4-tri-O-methyl-α-D-glucopyranosyl-(1→4)-O-2,3-di-O-methyl-β-D-glucopyranosyluronic acid-(1→4)-O-α-D-glucopyranosyl-(1→4)-O-2,3-di-O-methyl-α-L-idopyranosyluronic acid-(1→4)-O-α-D-glucopyranose, referred to hereinbelow as the compound of formula (I), corresponds to the following formula:
The compound of formula (I) in crystalline form according to the invention has a powder X-ray diffractogram whose characteristic lines are approximately at 12.009; 7.703; 7.300; 7.129; 5.838; 4.665; 4.476 and 3.785 angströms (interplanar distances). It also has a melting point of about 203° C. (203° C.±1° C.).
EXAMPLE 1 Preparation of the Compound of Formula (I) in Crystalline Form (Scheme 1)
Methyl O-2,3,4-tri-O-methyl-α-D-glucopyranosyl-(1→4)-O-2,3-di-O-methyl-β-D-glucopyranosyluronic acid-(1→4)-O-α-D-glucopyranosyl-(1→4)-O-2,3-di-O-methyl-α-L-idopyranosyluronic acid-(1→4)-O-α-D-glucopyranose, referred to hereinbelow as the compound of formula (I)
1.1: Preparation of the Compound of Formula (I′)
The compound of formula (I″) is obtained, for example, according to the teaching of patent EP 0 529 715 B1 or of the articles “Bioorg. Med. Chem.” (1994, Vol. 2, No. 11, pp. 1267-1280), “Bioorg. Med. Chem. Letters” (1992, Vol. 2, No. 9, pp. 905-910) or “Magnetic Resonance in Chemistry” (2001, Vol. 39, pp. 288-293). The compound of formula (I″) (5 g, 3.06 mmol) is dissolved in acetonitrile (10 mL). Deionized water (12.2 mL) and aqueous 30% sodium hydroxide solution (4.1 g) are then added. The mixture is heated to 40° C. and maintained at this temperature for 5 hours. The reaction medium is then cooled to 20° C. and acidified to pH 6.25 with aqueous 1N hydrochloric acid solution (about 17.7 g) before extraction with MTBE of certain impurities, the saponified product remaining in the aqueous phase. The residual acetonitrile, contained in the aqueous phase, is then removed by concentration, followed by diluting with deionized water (125 mL). The saponified product is finally precipitated at pH 1.5 by adding aqueous 1N hydrochloric acid solution (about 17.6 g) at 20° C. The suspension is maintained for 4 hours at 20° C. before filtration. The wet solid is finally dried in a vacuum oven at 30° C. to give 2.93 g (93.6%) of compound of formula (I).
NMR (anomeric protons of the saccharide units D, E, F, G, H): 5.79, 5.14, 5.55, 5.92, 4.94 ppm.
1.2 Preparation of the Crude Compound of Formula (I)
The compound of formula (I′) obtained after the preceding step is dissolved in tetrahydrofuran (18 mL). Palladium-on-charcoal (0.3 g) is added. The reaction medium is hydrogenated at 0.3 bar of hydrogen (relative pressure) for 4 hours. After filtering and evaporating, 2.12 g (99%) of the crude compound of formula (I) are obtained.
1.3: Preparation of the Compound of Formula (I) in Crystalline Form Using an Isopropanol/MTBE Mixture
The crude hydrogenated product obtained after the preceding step is dissolved in isopropanol (13 mL) at 65° C., and then crystallized at room temperature. The suspension is then cooled to 40° C., followed by addition of MTBE (13 mL), and is then cooled slowly to 10° C. After maintenance at 10° C. for 2 hours, the crystalline hydrogenated product is filtered off, washed and dried. 1.66 g of the compound of formula (I) in crystalline form are thus obtained, in the form of a cream-white powder. The reaction yield for the production of the compound of formula (I) in crystalline form, from the compound of formula (I′), is 92.5%. When expressed relative to the starting compound (I″), the reaction yield for the production of the compound of formula (I) in crystalline form is 86.6%.
NMR (anomeric protons of the saccharide units D, E, F, G, H) of the compound of formula (I) in crystalline form: 5.77, 5.11, 5.51, 5.84, 5.01 ppm.
1.4: Preparation of the Compound of Formula (I) in Crystalline Form Using Isopropanol
The crude hydrogenated product obtained after step 1.2 is dissolved in isopropanol (5 volumes) at 75° C. The medium is then cooled slowly until crystals appear, according to the known standard techniques for crystallization. The process is performed, for example, by a first step of cooling at 65° C. for 1 hour, and than a second step of cooling to a final temperature of 25° C. over 4 hours or of 5° C. over 6 hours, and finally maintenance at this final temperature for 30 minutes. The suspension is then filtered and rinsed with isopropanol (2×0.1 V) and compound (I) is isolated in the form of white crystals, which appear under a microscope in the form of needles. The 1H NMR analysis of these crystals is identical to that described after step 1.3 above.
EXAMPLE 4 Preparation of Idraparinux from the Compound of Formula (I) in Crystalline Form (Scheme 2)
The preparation of idraparinux (II) from the compound of formula (I) is summarized in Scheme 2.
The compound of formula (I) in crystalline form, as obtained according to Example 1.3, is dissolved in N,N′-dimethylformamide (6.6 mL) and then heated to 30° C. Under an inert atmosphere, 3.8 g of pyridine-sulfur trioxide complex are added slowly, followed by maintenance at 30° C. for 4 hours. The reaction medium is then poured into aqueous 23.8% sodium hydrogen carbonate solution (16.3 g) maintained at a maximum of 25° C., to obtain the compound of formula (II). The reaction medium is kept stirring for hours. The solution of sulfated product is then poured onto an MTBE/isopropanol/ethanol mixture (171 mL/70 mL/70 mL). Precipitation of the product is observed, and, after filtering off, washing and drying the cake, 4.99 g (96.8%) of compound of formula (II) are obtained, and are then purified by anion-exchange chromatography according to the usual techniques.
NMR (anomeric protons of the saccharide units D, E, F, G, H) of the compound of formula (II): 5.48, 4.68, 5.44, 5.08, 5.18 ppm.
It thus appears that the process according to the invention makes it possible to obtain idraparinux (compound of formula (II)) in a chemical yield of about 84% (precisely 83.8% according to the protocols described above) starting from the compound of formula (I″), i.e. a gain in yield of about 30% relative to the process described in patent EP 0 529 715 B1.
………………..
methyl O-2,3,4-tri-O-methyl-6-O-sulfo-α-D-glucopyranosyl-(1→4)-O-2,3-di-O-methyl-α-L-idopyranuronosyl-(1→4)-O-2,3,6-tri-O-sulfo-α-D-glucopyranosyl-(1→4)-O-2,3-O-di-methyl-α-L-idopyranuronosyl-(1→4)-O-2,3,6-tri-O-sulfo-α-D-glucopyranoside nonakis sodium salt. [α]D²⁰ = +46.2° (c=1; water). Anomeric protons chemical shifts: 5.43; 5.37; 5.16; 5.09; and 5.09 ppm.
WAS PREPARED AS PER
- Example 3
methyl O-4-O-(4-sulfoaminophenyl)-2,3,6-tri-O-sulfo-α-D-glucopyranosyl-(1→4)-O-3-O-methyl-2-O-sulfo-α-L-idopyranuronosyl-(1→4)-O-2,3,6-tri-O-sulfo-α-D-glucopyranoside nonakis sodium salt.
NOTE THIS IS ANALOGOUS PROCEDURE AND NOT SIMILAR
- Methyl O-4-O-(4-nitrophenyl)-6-O-acetyl-2,3-O-di-phenylmethyl-α-D-glucopyranosyl-(1→4)-O-(methyl 3-O-methyl-2-O-acetyl-α-L-idopyranosyluronate)-(1→4)-O-2,3,6-tri-O-acetyl-α-D-glucopyranoside (100 mg, 0.09 mmol), obtained by the known imidate coupling of the trichloroacetimidate of O-4-O-(4-nitrophenyl)-6-O-acetyl-2,3-O-di-phenylmethyl-α-D-glucopyranoside and methyl O-(methyl 3-O-methyl-2-O-acetyl-α-L-idopyranosyluronate)-(1→4)-O- 2,3,6-tri-O-acetyl-α-D-glucopyranoside, was dissolved in tetrahydrofuran (9 ml) and cooled to -5 °C. At this temperature a 30% aq. solution of hydrogen peroxide (4.5 ml) was added to the reaction mixture, and after 10 min a 1.25 M lithium hydroxide solution (4.7 ml) was added. The mixture was stirred for 1 h at -5 °C, after which time the temperature was raised to 0 °C and the mixture was stirred overnight. The reaction mixture was acidified with 6N hydrogen chloride at 0 °C to pH 1.5, after which the saponified compound was extracted with ethyl acetate. The organic layers were pooled, dried over magnesium sulfate, and evaporated to give 63 mg (84%) of methyl O-4-O-(4-nitrophenyl)-2,3-O-di-phenylmethy1-α-D-glucopyranosyl-(1→4)-O-3-O-methyl-α-L-idopyranuronosyl-(1→4)-O-α-D-glucopyranoside, which was dissolved in methanol (8 ml). 10% Pd on charcoal (63 mg) was added and the mixture hydrogenolyzed overnight. After filtration and evaporation 27 mg (50%) of methyl O-4-O-(4-aminophenyl)-α-D-glucopyranosyl-(1→4)-O-3-O-methyl-α-L-idopyranuronosyl-(1→4)-O-α-D-glucopyranoside were obtained.
13 mg of methyl O-4-O-(4-aminophenyl)-O-α-D-glucopyranosyl-(1→4)-O-3-O-methyl-α-L-idopyranuronosyl-(1→4)-O-α-D-glucopyranoside were dissolved in 2 ml of dry N,N-dimethylformamide, and under an atmosphere of nitrogen 148 mg of triethylamine sulfurtrioxide complex were added. The mixture was stirred overnight at 50 °C, after which an aq. solution of sodium hydrogen carbonate was added under ice cooling. The mixture was stirred for 1 h at room temperature, concentrated to a small volume and desalted on a Sephadex G-10 column with water. The crude product obtained was purified by HPLC using a Mono-Q anion exchange column to give 11 mg (37%) of methyl O-4-O-(4-sulfoaminophenyl)-2,3,6-tri-O-sulfo-α-D-glucopyranosyl-(1→4)-O-3-O-methyl-2-O-sulfo-α-L-idopyranuronosyl-(1→4)-O-2,3,6-tri-O-sulfo-α-D-glucopyranoside nonakis sodium salt. [α]D²⁰ = +52.2° (c=0.67; water). Anomeric protons chemical shifts: 5.5; 5.17; and 5.15 ppm.
………………………………..
BMCL Volume 19, Issue 14, 15 July 2009, Pages 3875–3879


Final elaboration of the pentasaccharide 1. Reagents and conditions: (a) TMSOTf, Et2O, 4 Å MS, rt, 66% (28α), 15% (28β); (b) CAN, CH3CN, toluene, H2O, rt, 72%; (c) CCl3CN, DBU, CH2Cl2, rt, 98%; (d) TMSOTf, 4 Å MS, CH2Cl2, rt, 51% (73% based on recovery of 4); (e) Pd/C (10%), H2, t-BuOH, H2O, rt; (f) SO3·Et3N, DMF, 50 °C, 93% (2 steps).
The final elaboration of the pentasaccharide 1 was illustrated in IN ABOVE SCHEME Coupling of the glucopyranosyl trichloroacetimidate 6 with disaccharide acceptor 5 in the presence of trimethylsilyl trifluoromethylsulfonate and powdered 4 Å molecular sieves at room temperature in diethyl ether afforded the desired α-coupled trisaccharide 28α in a yield of 66%, together with 15% of the separable β-coupled product 28β. The anomeric 4-methoxyphenyl group in trisaccharide 28α was removed with CAN, and the resulting lactol was readily converted into the trisaccharide trichloroacetimidate 3. Coupling of donor 3 with the disaccharide acceptor 4 in the presence of trimethylsilyl trifluoromethylsulfonate and powdered 4 Å molecular sieves at room temperature in dichloromethane afforded the fully protected pentasaccharide 2 in 51% yield (73% based on recovery of 4). Finally, pentasaccharide 2 was subject to hydrogenolysis of the benzyl protecting groups. The highly polar product without purification was O-sulfated directly with triethylamine-sulfur trioxide complex to afford the sulfated pentasaccharide 1 in an excellent yield of 93% (for two steps).
Summarizing, the potent anti-thromboembolic pentasaccharide Idraparinux (1) was synthesized in total 51 steps and in 4% overall yield from d-glucose and methyl α-d-glucopyranoside.18 The synthetic route is convergent with a linear sequence of 27 steps, and the transformations are scalable. The 4-methoxyphenol glycoside intermediates are easy to be purified by crystallization.
1H NMR (400 MHz, D2O) δ 3.27 (t, J = 8.4 Hz, 1H), 3.30–3.38 (m, 2H), 3.47 (s, 3H), 3.53 (s, 3H), 3.56 (s, 6H), 3.58 (s, 3H), 3.62 (s, 3H), 3.63 (s, 3H), 3.64 (s, 6H), 3.75 (d, J = 10.0 Hz, 1H), 3.83–3.97 (m, 4H), 3.98 (t, J = 8.8 Hz, 1H), 4.06–4.18 (m, 3H), 4.19–4.45 (m, 8H), 4.56 (br t, J = 9.6 Hz, 1H), 4.65 (t, J = 9.2 Hz, 1H), 4.66 (d, J = 7.6 Hz, 1H), 5.00 (br s, 1H), 5.11 (br s, 1H), 5.17 (d, J = 3.6 Hz, 1H), 5.43 (d,J = 3.2 Hz, 1H), 5.47 (d, J = 4.0 Hz, 1H);
ESI-MS m/z 774.1 [M−8Na+6H]2−, 763.0; [M−9Na+7H]2−, 508.5 [M−9Na+6H]3−.
………………….
The process of preparation of Idraparinux having the following formula:
may comprise the following steps :
1 ) preparation a compound of formula (IXB)
(IXB) wherein Ra is methyl, Rb is methyl, Rc is methyl, T-i is benzyl, T2 is benzyl, T3 is benzyl and T is methyl, by the process according to the invention;
2) epimerisation of the disaccharide (IXB) so as to form disaccharide D of formula :
3) protection of the 4′-OH of D with a levulinoyl ester;
4) acetolysis of the disaccharide resulting from step 3), followed by preparation of the corresponding imidate;
5) coupling the disaccharide imidate resulting from step 4) with (IXB) obtainable by the process of the invention, wherein Ra is methyl, Rb is methyl, Rc is methyl, T-i is benzyl, T2 is benzyl, T3 is benzyl and T is methyl, to obtain a tetrasaccharide;
6) coupling the fully protected tetrasaccharide with a monosaccharide glycosyl imidate;
7) deprotection of the protecting groups by the successive saponification and hydrogenolysis;
8) sulfation of the hydroxyl groups.
In one embodiment, the present invention concerns a process of preparation of Idraparinux:
said process comprising the following steps:
preparation of a compound of formula (VI) such as defined above, from a compound of formula (V) such as defined above; preparation of a compound of formula (VII) such as defined above, from a compound of formula (VI) such as defined above;
preparation of a compound of formula (VIII) such as defined above, from a compound of formula (VII) such as defined above;
- preparation of a compound of formula (IX) such as defined above, from a compound of formula (VIII) such as defined above;
wherein in compounds of formulae (V), (VI), (VII), (VIII) and (IX), R-i , R2, R3 and X are as defined above, Ra is methyl, Rb is methyl, Rc is methyl, Rd is methyl and R’ is the monosaccharide of formula :
wherein T-i is benzyl, T2 is benzyl, T3 is benzyl and T is methyl. The inventors advantageously found that the process of preparation of Idraparinux comprising the decarboxylation/intramolecular cyclisation tandem reaction, which allows the inversion of configuration of C5 carbon of the compound of formula (VI), is more efficient than the processes previously described in the literature. Indeed, the process according to the invention allows advantageously a significant decrease of the number of steps and thus an improvement of the overall yield. Thus, the process of preparation of Idraparinux may be carried out in industrial scales. The inventors found an efficient process of preparation of Idraparinux.
According to another object, the present invention concerns the use of compounds of formulae (V), (VI), (VI I), (VIII) and (IX), as intermediates for the preparation of Idraparinux. In particular, the present invention concerns the use of a compounds of formulae (VB), (VI B), (VI IB), (VI I IB) and (IXB), as intermediates for the preparation of Idraparinux The invention is further illustrated but not restricted by the description in the following examples. Example 1 :
Preparation of Methyl-4,6-0-benzylidene-a-D-glucopyranoside (la)
CHC13)
Tf = 166-167°C (litt. 165-166°C) To a solution of benzaldehyde (400 mL, 3.94 mol, 5.9 eq.) was added zinc chloride (100.3 g, 0.74 mol, 1 .1 eq.) under vigorous stirring. After homogenization of the solution methyl- a-D-glucopyranoside (129.6 g, 0.67 mol, 1.0 eq.) was added portionwise. After 16 hours stirring at room temperature the reaction mixture was diluted with diethyl ether (100 mL). The mixture was then poured dropwise and under vigorous stirring in a solution containing ice water (1 .5 L) and hexane (350 mL). The precipitate was filtered, washed with diethyl ether (3 x 300 mL) and dried under vacuum over KOH. The product was then recrystallised from CH2CI2 (720 mL) and washed with a Et20/CH2CI2 solution (75:25, 2 x 200 mL). The filtrate was repeatedly recrystallised five times from CH2CI2 to afford compound la as white crystals (136.97 g, 0.49 mol, 72%).
1H NMR (CDCI3, 250 MHz): δ 2.35 (d, JCH-OH = 9.2 Hz, 1 H, OH), 2.83 (d, JCH-OH = 2.2 Hz, 1 H, OH), 3.46 (s, 3H, -OCH3), 3.43-3.46 (m, 1 H, H-4), 3.63 (td, JCH-OH = ^2,3 = 9.2 Hz, J1 2 = 3.9 Hz, 1 H, H-2), 3.70-3.81 (m, 2H, H-5, H-6), 3.93 (td, J = 9.2 Hz, JCH-OH = 2.2 Hz, 1 H, H- 3), 4.29 (m, 1 H, H-6 ), 4.79 (d, J1i2 = 3.9 Hz, 1 H, H-1 ), 5.54 (s, 1 H, Ph-CH), 7.35-7.38 (m, 3H, HAr), 7.47-7.51 (m, 2H, HAr).
13C NMR (CDCI3, 62.9 MHz): δ 55.6 (-OCH3), 62.5 (C-5), 69.0 (C-6), 71.1 (C-3), 72.9 (C- 2), 81 .0 (C-4), 99.9 (C-1 ), 102.0 (Ph-CH), 126.4, 128.5, 129.4, 137.1 (6xCAr). IR (film) v (cm“1): 3369 (O-H). Preparation of Methyl-2,3-di-0-methyl-4,6-0-benzylidene-a-D-glucopyranoside (Ma)
13)
°C)
To a solution of compound la (47.60 g, 0.17 mol, 1.0 eq.) in anhydrous THF (750 mL) under an argon atmosphere and cooled to 0°C, was added portionwise sodium hydride (60%, 16.93 g, 0.42 mol, 2.5 eq.). After 20 minutes methyl iodide was added dropwise (30 mL, 0.48 mol, 2.8 eq.) and the reaction mixture was allowed to reach room temperature. After 16 hours, methanol was added portionwise (75 mL) and the solution was stirred for another 15 minutes before being concentrated. The resulting residue was dissolved in EtOAc (400 mL) and washed with water (2 x 250 mL). The organic layer was dried (MgS04), filtered and concentrated. The resulting solid was dissolved in diethyl ether (1000 mL), hexane was added (400 mL) and the solvent was partially evaporated at low temperature. The crystals obtained were washed with hexane and the filtrate once again partially evaporated, filtered and the precipitate washed with hexane. The combined precipitates afforded compound Ma as white crystals (46.10 g, 0.15 mol, 88%).
1H NMR (CDCI3, 250 MHz): δ 3.30 (dd, J2-3 = 9.1 Hz, J1-2 = 3.7 Hz, 1 H, H-2), 3.44 (s, 3H, – OCH3), 3.49-3.87 (m, 4H, H-4, H-5, H-6, H-3), 3.55 (s, 3H, -OCH3), 3.64 (s, 3H, -OCH3), 4.28 (dd, J6-6. = 9.1 Hz, J5-6 = 3.7 Hz, 1 H, H-6 ), 4.85 (d, J1-2 = 3.7 Hz, 1 H, H-1 ), 5.54 (s, 1 H, Ph-CH), 7.36-7.41 (m, 3H, HAr), 7.48-7.52 (m, 2H, HAr).
13C NMR (CDCI3, 62.9 MHz): δ 55.4, 59.5, 61.1 (3x-OCH3), 62.3 (C-5), 69.1 (C-6), 79.9 (C-3), 81 .5 (C-4), 82.2 (C-2), 98.5 (C-1 ), 101.4 (Ph-CH), 126.2, 128.3, 129.0, 137.4 (6xCAr).
Preparation of Methyl-2,3-di-0-methyl-a-D-glucopyranoside (Ilia)
13)
To a suspension of compound Ma (10.33 g, 33.29 mmol, 1.0 eq.) in methanol (150 mL) was added para-toluenesulfonic acid monohydrate (322 mg, 1 .69 mmol, 0.05 eq.). After 4 hours stirring at room temperature, sodium carbonate (300 mg) was added and the reaction mixture was stirred an additional 15 minutes before filtration through a pad of Celite®. Then the filtrate was concentrated and the residue obtained was dissolved in a mixture of distilled water/diethyl ether (3:1 , 150 mL). The organic layer was extracted with water (2 x 50 mL) then the combined aqueous phases were concentrated and dried one night under vacuum over KOH. The resulting residue was recrystallised from toluene using petroleum ether as a co-solvent. The crystals obtained were washed with hexane and dried under vacuum over KOH to obtain compound Ilia as white crystals (6.63 g, 29.83 mmol, 90%).
1H NMR (DMSO, 400 MHz): δ 3.03 (dd, J2-3 = 9.3 Hz, J1-2 = 3.5 Hz, 1 H, H-2), 3.12-3.20 (m, 2H, H-3, H-4), 3.27 (s, 3H, -OCH3), 3.32 (s, 3H, -OCH3), 3.30-3.33 (m, 1 H, H-5), 3.44 (s, 3H, -OCH3), 3.40-3.46 (m, 1 H, H-6), 3.62 (ddd, J6-6‘ = 1 1.6 Hz, JCH-OH = 5.7 Hz, J5-6 = 1 ,9 Hz, 1 H, H-6′), 4.52 (t, J = 5.7 Hz, 1 H, OH), 4.78 (d, Ji-2 = 3.5 Hz, 1 H, H-1 ), 5.09 (d,
13C NMR (DMSO, 100.6 MHz): δ 54.1 , 57.4, 60.0 (3x-OCH3), 60.6 (C-6), 69.5 (C-3), 72.5 (C-5), 80.9 (C-2), 82.8 (C-4), 96.4 (C-1 ).
IR (film) v (cm“1): 3419 (O-H).
Preparation of Methyl methyl-2,3-di-0-methyl-a-D-glucopyranosiduronate (IVa)
C13)
To a solution of compound Ilia (500 mg, 2.25 mmol, 1.0 eq.) in distilled water (15 mL) were successively added NaBr (50 mg, 0.49 mmol, 0.2 eq.) and TEMPO (7 mg, 0.05 mmol, 0.02 eq.). The reaction mixture was cooled with the aid of an ice bath then a solution of NaOCI (13% v/v, 5.2 mL, 9.1 mmol, 4.0 eq.) was added. After 5 hours stirring at 0°C ethanol was added (96% v/v, 8 mL), then the pH was reduced to 2-3 by addition of HCI (10 % v/v). The solvent was evaporated and the residue obtained was suspended in methanol, filtered in order to remove the remaining salts and washed several times with dichloromethane and methanol. The filtrate was concentrated then dissolved, under an argon atmosphere, in dry methanol (40 mL). para-toluenesulfonic acid (85 mg, 0.45 mmol, 0.2 eq.) was added then the reaction mixture was heated under reflux overnight. The solvent was evaporated and the residue obtained was dissolved in EtOAc (60 mL). The organic layer was washed with a 5% aqueous NaHC03 solution (2 χ 20 mL) and with brine (1 χ 20 mL). The aqueous phase was extracted with dichloromethane (3 χ 20 mL). The combined organics were dried (MgS04), filtered and evaporated. Column chromatography (hexane/ethyl acetate 50:50) gave compound IVa as a colourless oil (503 mg, 2.00 mmol, 89%).
1H NMR (CDCIs, 400 MHz): δ 3.10 (d, JCH-OH = 3.0 Hz, 1 H, OH), 3.26 (dd, J2-3 = 9.3 Hz, J1 -2 = 3.4 Hz, 1 H, H-2), 3.47 (s, 3H, -OCH3), 3.49-3.52 (m, 1 H, H-3), 3.50 (s, 3H, -OCH3), 3.62 (s, 3H, -OCH3), 3.74 (td, J = 9.5 Hz, JCH-OH = 3.0 Hz, 1 H, H-4), 3.82 (s, 3H, -OCH3), 4.14 (d, J4.5 = 9.6 Hz, 1 H, H-5), 4.91 (d, Ji-2 = 3.4 Hz, 1 H, H-1 ).
13C NMR (CDCI3, 100.6 MHz): δ 52.9, 56.0, 59.1 , 61 .3 (4x-OCH3), 70.6 (C-5), 71.7 (C-4), 80.9 (C-2), 81.8 (C-3), 98.1 (C-1 ), 170.9 (C=0).
IR (film) v (cm“1): 3475 (O-H), 1750 (C=0).
Preparation of Methyl methyl^-O-il’-ethoxy^’-propyn-l’-ylJ^.S-di-O-methyl-a-D- glucopyranosiduronate (Va)
To a solution of compound IVa (4.56 g, 18.21 mmol, 1.0 eq.) in chloroform (stabilised with amylene, 200 mL) were added, under an argon atmosphere, P205 (5.31 g, 36.29 mmol, 2.0 eq.) and propargylaldehyde diethylacetal (5.2 mL, 36.27 mmol, 2.0 eq.), then the reaction mixture was heated at 60°C. After 4 hours stirring, the reaction mixture was filtered through a pad of Celite® then the solvent was removed under vacuum. The crude mixture was suspended in EtOAc (300 mL), washed with a 5% NaHC03 aqueous solution (1 x 30 mL) and brine (1 x 30 ml_). The organic layer was dried (MgS04), filtered, and evaporated. Column chromatography (gradient hexane/ethyl acetate 80:20 – 20:80) afforded compound Va as a colourless oil (4.07 g, 12.24 mmol, 67%) in a diastereomeric mixture (64:36) (the relative composition of the mixture was determined by 1H NMR from integrations of protons EtO-CH), along with some unreacted compound IVa (1.17 g, 4.68 mmol, 26%).
1H NMR (CDCI3, 400 MHz): δ 1.18-1.25 (m, 3H, -OCH2CH3) (diastereomeric mixture), 2.56 (m, 1 H, H-C≡C-) (mixture), 3.26-3.31 (m, 1 H, H-2) (mixture), 3.43 (s, 3H, -OCH3) (major), 3.44 (s, 3H, -OCH3) (minor), 3.50 (s, 3H, -OCH3) (mixture), 3.59 (s, 3H, -OCH3) (minor), 3.62 (s, 3H, -OCH3) (major), 3.47-3.62 (m, 2H, H-3, -OCHaHbCH3) (mixture), 3.65-3.73 (m, 1 H, -OCHaHbCH3) (mixture), 3.78 (s, 3H, -OCH3) (major), 3.80 (s, 3H, -OCH3) (minor), 3.78-3.86 (m, 1 H, H-4) (mixture), 4.15 (d, J4-5 = 10.0 Hz, 1 H, H-5) (major), 4.18 (d, J4-5 = 10.0 Hz, 1 H, H-5) (minor), 4.86-4.88 (m, 1 H, H-1 ) (mixture), 5.35 (d, J = 1.7 Hz, 1 H, EtO- CH) (minor), 5.58 (d, J = 1.7 Hz, 1 H, EtO-CH) (major).
13C NMR (CDCI3, 100.6 MHz): δ 15.0 (-OCH2CH3) (mixture), 52.6 (-OCH3) (major), 52.7 (- OCH3) (minor), 55.8 (-OCH3) (mixture), 59.2 (-OCH3) (major), 59.3 (-OCH3) (minor), 60.4 (-OCH2CH3) (major), 61 .3 (-OCH2CH3) (minor), 61.4 (-OCH3) (mixture), 70.1 (C-5) (minor), 70.2 (C-5) (major), 74.0 (H-C≡C-) (major), 74.2 (H-C≡C-) (minor), 76.4 (C-4) (minor), 76.7 (C-4) (major), 78.6 (H-C≡C-) (minor), 78.9 (H-C≡C-) (major), 81 .5 (C-2) (major), 81 .8 (C-2) (minor), 81.9 (C-3) (minor), 82.9 (C-3) (major), 92.6 (EtO-CH) (mixture), 97.9 (C-1 ) (minor), 98.0 (C-1 ) (major), 169.6 (C=0) (major), 169.9 (C=0) (minor). Elemental analysis: Calculated: C: 54.21 ; H: 7.28. Found: C: 54.17 ; H: 7.13.
ESI-MS (pos. mode): m/z = 355 [M+Na]+.
IR (film) v (cm“1): 1752 (C=0), 3266 (≡C-H). Preparation of 4-0-(1′-ethoxy-2′-propyn-1,-yl)-1 ,2,3-tri-0-methyl-a-D-gluco- pyranosiduronic acid (Via)
To a solution of compound Va (1.12 g, 3.37 mmol, 1.0 eq.) in EtOH/H20 (3:1 , 100 mL) was added sodium hydroxide (156 mg, 3.90 mmol, 1 .3 eq.). After 5 hours stirring at room temperature the solvent was evaporated. The residue obtained was dissolved in water (50 mL). The pH of the aqueous layer was reduced to 2-3 with a 5% citric acid aqueous solution, then the layer was saturated with sodium chloride before extraction with dichloromethane (10 x 20 mL). If necessary the pH was adjusted by addition of more citric acid aqueous solution. The combined organics were dried (MgS04), filtered and removed under vacuum. Compound Via was obtained without further purification as a colourless oil (1.020 g, 3.20 mmol, 95%), in a mixture of diastereomers (75:25) (the relative composition of the mixture was determined by 1H NMR from integrations of protons EtO-CH).
1H NMR (CDCIs, 400 MHz): δ 1.16-1.24 (m, 3H, -OCH2CH3) (diastereomeric mixture), 2.59 (d, J = 1 .6 Hz, 1 H, H-C≡C-) (major), 2.62 (s I, 1 H, H-C≡C-) (minor), 3.25-3.33 (m, 1 H, H- 2), 3.44 (s, 3H, -OCH3) (mixture), 3.51 (s, 3H, -OCH3) (mixture), 3.62 (s, 3H, -OCH3) (mixture), 3.54-3.62 (m, 2H, H-3, -OCHaHbCH3) (mixture), 3.68-3.77 (m, 1 H, -OCHaHbCH3) (mixture), 3.81-3.87 (m, 1 H, H-4) (mixture), 4.13-4.18 (m, 1 H, H-5) (mixture), 4.88-4.90 (m, 1 H, H-1 ), 5.45 (s I, 1 H, EtO-CH) (minor), 5.63 (d, J = 1.6 Hz, 1 H, EtO-CH) (major).
13C NMR (CDCI3, 100.6 MHz): δ 14.9 (-OCH2CH3) (mixture), 55.9 (-OCH3) (mixture), 59.2 (-OCH3) (minor), 59.3 (-OCH3) (major), 60.7 (-OCH2CH3) (mixture), 61 .2 (-OCH3) (minor), 61.3 (-OCH3) (major), 70.1 (C-5) (mixture), 74.3 (H-OC-) (major), 74.8 (H-OC-) (minor), 75.7 (C-4) (minor), 76.4 (C-4) (major), 78.5 (H-C≡C-) (minor), 78.8 (H-C≡C-) (major), 81.4 (C-2) (major), 81 .7 (C-2) (minor), 81 .8 (C-3) (minor), 82.9 (C-3) (major), 92.5 (EtO-CH) (mixture), 97.8 (C-1 ) (minor), 98.0 (C-1 ) (major), 173.8 (C=0) (major), 174.0 (C=0) (minor).
ESI-MS (pos. mode): m/z = 341 [M+Na]+. IR (film) v (cm“1): 1751 (C=0), 3268 (≡C-H).
Preparation of Methyl-4,7-anhydro-6-deoxy-6-methylene-7-ethoxy-2,3-di-0-methyl- a-L-/‘d -heptopyranoside (Vila)
To a solution of compound Via (1.89 g, 5.92 mmol, 1 .0 eq.) in anhydrous THF (40 mL) and cooled to 0°C, were added IBCF (0.84 mL, 6.48 mmol, 1.1 eq.) and N- methylmorpholine (0.72 mL, 6.55 mmol, 1.1 eq.). After 20 minutes stirring the flask was covered with aluminium foil, 2-mercaptopyridine /V-oxide sodium salt (1 .77 g, 1 1.80 mmol, 2.0 eq.) was added and the reaction mixture was stirred at ambient temperature. After 2 hours, anhydrous THF (80 mL) then ie f-butylthiol (0.28 mL, 2.61 mmol, 1.6 eq.) were added. The aluminium foil was removed and the reaction mixture was irradiated and heated 30 minutes with a UV lamp (300W). The thiol excess was neutralized with a NaOCI aqueous solution (13% v/v, 10 mL). The reaction mixture was concentrated then dissolved in EtOAc (100 mL), washed successively with a 5% NaHC03 aqueous solution (2 x 15 mL), a 5 % citric acid aqueous solution (1 x 15 mL) and brine (1 x 25 mL), then the aqueous layer was extracted with dichloromethane (2 x 20 mL). The combined organics were dried (MgS04), filtered and concentrated. Column chromatography (gradient dichloromethane/ethyl acetate 95:5 – 75:25) afforded compound Vila as a colourless oil (218 mg, 0.79 mmol, 48%), in a mixture of diastereomers (67:33) (the relative composition of the mixture was determined by 1H NMR from integrations of protons H-2).
1H NMR (CDCIs, 400 MHz): δ 1.23 (t, J = 7.1 Hz, 3H, -OCH2CH3) (diastereomeric mixture), 3.13 (dd, J2-3 = 9.6 Hz, J1-2 = 3.0 Hz, 1 H, H-2) (minor), 3.30 (dd, J2-3 = 5.0 Hz, J1-2 = 1.6 Hz, 1 H, H-2) (major), 3.41 (s, 3H, -OCH3) (minor), 3.47 (s, 3H, -OCH3) (major), 3.50 (s, 3H, -OCH3) (mixture), 3.53 (s, 3H, -OCH3) (major), 3.55-3.61 (m, 1 H, -OCHaHbCH3) (mixture), 3.72 (dd, J2-3 = 5.0 Hz, J3-4 = 2.8 Hz, 1 H, H-3) (major), 3.78-3.90 (m, 1 H, – OCHaHbCH3) (mixture), 3.93-4.07 (m, 2H, H-3, H-4) (mixture), 4.59 (d I, J4-5 = 4.0 Hz, 1 H, H-5) (major), 4.62 (d, J1-2 = 1.7 Hz, 1 H, H-1 ) (major), (td, J4-5 = 7.9 Hz, J = 2.6 Hz, 1 H, H- 5), (minor), 4.79 (d, J1-2 = 3.0 Hz, 1 H, H-1 ) (minor), 5.35-5.57 (m, 3H, H-7, -C=CH2) (mixture). 13C NMR (CDCI3, 100.6 MHz): δ 15.3 (-OCH2CH3) (minor), 15.4 (-OCH2CH3) (major), 56.5 (-OCH3) (minor), 56.8 (-OCH3) (major), 58.6 (-OCH3) (major), 59.1 (-OCH3) (minor), 59.9 (- OCH3) (major), 60.3 (-OCH3) (minor), 63.3 (-OCH2CH3) (minor), 63.8 (-OCH2CH3) (major), 74.2 (C-5) (minor), 74.7 (C-5) (major), 76.4 (C-3) (major), 77.0 (C-4) (major), 77.7 (C-2) (major), 79.0 (C-3) (minor), 79.7 (C-4) (minor), 80.1 (C-2) (minor), 99.3 (C-1 ) (major), 99.5 (C-1 ) (minor), 102.2 (C-7) (major), 103.0 (C-7) (minor), 1 1 1.6 (-C=CH2) (minor), 1 15.2 (- C=CH2) (major), 147.4 (C-6) (minor), 148.1 (C-6) (major).
ESI-MS (pos. mode): m/z = 297 [M+Na]+.
Preparation of Methyl-4,7-anhydro-7-ethoxy-2,3-di-0-methyl-a-L-/ o-hepto- pyranosid-6-ulose (Villa)
Through a solution of compound Vila (449 mg, 1 .64 mmol, 1.0 eq.) in anhydrous dichloromethane (10 ml_), under an argon atmosphere and cooled to -78°C, was bubbled ozone (0.2 L/min, 1 10 V). When the solution had turned dark blue, oxygen was bubbled through in order to remove the excess ozone. When the solution became colorless dimethylsulfide (5 drops) was added and the solution was brought to room temperature. After 1 h15 the reaction mixture was concentrated. Column chromatography (gradient dichloromethane/ethyl acetate 95:5 – 80:20) afforded compound Villa as a white solid (364 mg, 1.32 mmol, 80%), in a mixture of diastereomers (79:21 ) (the relative composition of the mixture was determined by 1H NMR from integrations of protons H-2).
1H NMR (CDCI3, 400 MHz): δ 1.24-1 .28 (m, 3H, -OCH2CH3) (diastereomeric mixture), 3.10 (dd, J2-3 = 10.2 Hz, J1-2 = 2.9 Hz, 1 H, H-2) (minor), 3.17 (dd, J2-3 = 9.4 Hz, J1-2 = 2.8 Hz, 1 H, H-2) (major), 3.38 (s, 3H, -OCH3) (major), 3.42 (s, 3H, -OCH3) (minor), 3.50 (s, 3H, – OCH3) (mixture), 3.63 (s, 3H, -OCH3) (major), 3.66 (s, 3H, -OCH3) (minor), 3.48-3.73 (m, 3H, H-3 major, -OCHaHbCH3 major, -OCHaHbCH3 minor), 3.77-3.95 (m, 2H, -OCHaHbCH3 minor, -OCHaHbCH3 major), 4.07 (dd, J2-3 = 10.2 Hz, J3-4 = 7.7 Hz, 1 H, H-3) (minor), 4.34 (d, J4-5 = 9.1 Hz, 1 H, H-5) (minor), 4.39-4.44 (m, 2H, H-4 minor, H-5 major), 4.50 (dd, J3-4 = 9.5 Hz, J4-5 = 6.2 Hz, 1 H, H-4) (major), 4.76 (d, Ji-2 = 2.8 Hz, 1 H, H-1 ) (major), 4.79 (d, J1-2 = 2.9 Hz, 1 H, H-1 ) (minor), 4.89 (d, J = 1 ,1 Hz, 1 H, H-7) (minor), 4.93 (s I, 1 H, H-7) (major).
13C NMR (CDCI3, 100.6 MHz): δ 15.1 (-OCH2CH3) (minor), 15.2 (-OCH2CH3) (major), 56.7 (-OCH3) (minor), 57.2 (-OCH3) (major), 59.3 (-OCH3) (mixture), 59.8 (-OCH3) (major), 60.6 (-OCH3) (minor), 65.0 (-OCH2CH3) (minor), 65.5 (-OCH2CH3) (major), 70.2 (C-5) (major), 72.4 (C-5) (minor), 75.9 (C-4) (major), 79.2 (C-4) (minor), 79.4 (C-3) (major), 79.8 (C-2 major, C-3 minor), 80.2 (C-2) (minor), 96.1 (C-7) (major), 97.2 (C-7) (minor), 98.7 (C-1 ) (major), 99.0 (C-1 ) (minor), 205.3 (C-6) (minor), 205.6 (C-6) (major).
IR (film) v (cm“1): 1783 (C=0).
ESI-MS (pos. mode): m/z = 299 [M+Na]+, 331 [M+Na+MeOH]+.
Preparation of Methyl methyl-2,3-di-0-methyl-a-L-idopyranosiduronate (IXa)
; CHCI3)
To a solution of compound Villa (50 mg, 0.18 mmol, 1 .0 eq.) in dichloromethane (3 mL), under an argon atmosphere and cooled to 0°C, were added m-CPBA (77%, 120 mg, 0.54 mmol, 3.0 eq.) and NaHC03 (20 mg, 0.23 mmol, 1 .3 eq.). After 3 hours stirring the solvent was removed under vacuum. The resulting residue was dissolved in EtOAc (30 mL), extracted with distilled water (2 x 10 mL), and the aqueous phase was concentrated. The crude mixture was dissoved in methanol (10 mL), para-toluenesulfonic acid monohydrate was added (4 mg, 0.02 mmol, 0.1 eq.) then the reaction mixture was heated to reflux and the reaction monitored by 1H NMR in deuterated methanol. After 8 hours the solvent was evaporated. The residue obtained was dissolved in DMF (5 mL) then triethylamine (28 μί, 0.20 mmol, 1 .1 eq.) and methyl iodide (56 μί, 0.90 mmol, 5 eq.) were added. After 3h30 the reaction mixture was concentrated, dissolved in EtOAc (30 mL) and the organic phase was washed with a 5% NaHC03 aqueous solution (2 x 10 mL), a 5% citric acid aqueous solution (2 x 10 mL) and brine (1 x 10 mL). The aqueous phase was extracted with dichloromethane (5 x 10 mL) and the combined organics were dried (MgS04), filtered and concentrated. Column chromatography (dichloromethane/ethyl acetate 85:15) afforded compound Xla as a colourless oil (25 mg, 0.10 mmol, 56%). 1H NMR (CDCI3, 400 MHz): δ 3.41 (d I, J2-3 = 3.5 Hz, 1 H, H-2), 3.47 (s, 3H, -OCH3), 3.56 (s, 3H, -OCH3), 3.57 (s, 3H, -OCH3), 3.69 (t, J2-3 = J3-4 = 3.5 Hz, 1 H, H-3), 3.75-3.78 (m, 1 H, OH), 3.80 (s, 3H, -OCH3), 3.97 (m, 1 H, H-4), 4.42 (d, J4-5 = 1 .6 Hz, 1 H, H-5), 4.61 (d,
13C NMR (CDCI3, 100.6 MHz): δ 52.4, 57.5, 58.4, 60.8 (4x-OCH3), 67.7 (C-4), 74.8 (C-5), 77.2 (C-2), 77.5 (C-3), 100.9 (C-1 ), 169.6 (C=0).
Elemental analysis: Calculated: C: 48.00 ; H: 7.25. Found: C: 47.62 ; H: 7.15.
ESI-MS (pos. mode): m/z = 272 [M+Na]+.
IR (film) v (cm“1): 3491 (O-H), 1765 (C=0). Example 2 :
Preparation of Methyl-2,3,6-tri-0-benzyl-4-0(2′,3′-di-0-methyl-p-D-glucopyranosyl- uronate)-a-D-glucopyranoside (IVb)
To a solution of the co
Me
(3.279 g, 5.00 mmol, 1.0 eq.) in a water/acetonitrile mixture (1 :1 , 300 mL) were added NaBr (105 mg, 1 .02 mmol, 0.2 eq.) and TEMPO (33 mg, 0.21 mmol, 0.04 eq.). The reaction mixture was cooled with the aid of an ice bath then a solution of NaOCI (13% v/v, 1 1.5 mL, 20.08 mmol, 4.0 eq.) was added. After 3 hours stirring at 0°C, NaOCI was added anew (13% v/v, 1 1 .5 mL, 20.08 mmol, 4.0 eq.). After two more hours ethanol was added (96% v/v, 20 mL), then the pH was reduced to 2-3 by addition of HCI (10% v/v). The solvent was evaporated and the residue obtained was suspended in DMF (40 mL) then triethylamine (2.8 mL, 2.032 g, 20.0 mmol, 4.0 eq.) and methyl iodide (6.2 mL, 14.136 g, 99.6 mmol, 20.0 eq.) were added. After 4 hours stirring at room temperature the solvent was evaporated and the residue obtained was dissolved in EtOAc (200 mL). The organic layer was washed with a 5% citric acid aqueous solution (1 χ 20 mL) and brine (1 χ 20 mL). The aqueous layer was extracted with dichloromethane (2 χ 20 mL). The combined organics were dried (MgS04), filtered and evaporated. The residue obtained was dissolved in DMF (20 mL), then triethylamine (1.4 mL, 1 .016 g, 10.0 mmol, 2.0 eq.) and methyl iodide (3.1 mL, 7.068 g, 49.8 mmol, 10.0 eq) were added. After 60 hours stirring at room temperature the solvent was evaporated and the residue obtained was dissolved in EtOAc (200 mL). The organic layer was washed with a 5% citric acid aqueous solution (2 x 20 mL) and brine (1 χ 20 mL). The organic layer was dried (MgS04), filtered and evaporated. Column chromatography (gradient hexane/ethyl acetate 80:20 – 50:50) gave compound IVb as a colourless oil which was dissolved in a diethyl ether/hexane mixture and evaporated at room temperature to afford a white solid (2.500 g, 3.66 mmol, 73%).
1H NMR (CDCI3, 400 MHz): δ 2.92 (d, 1 H), 2.93-2.98 (m, 2H), 3.41 (s, 3H), 3.48-3.76 (m, 5H), 3.51 (s, 3H), 3.60 (s, 3H), 3.63 (s, 3H), 3.87-3.98 (m, 3H), 4.36-4.41 (m, 1 H), 4.49- 5.08 (m, 6H), 4.61 (d, 1 H), 7.24-7.42 (m, 15H).
Elemental analysis: Calculated: C: 65.09 ; H: 6.79. Found: C: 65.29 ; H: 6.96.
ESI-MS (pos. mode): m/z = 705 [M+Na]+.
Preparation of Methyl-2,3,6-tri-0-benzyl-4-0(4′-0-(1 “-ethoxy-2″-propyn-1 “-yl)-2′,3′- di-0-methyl-p-D-glucopyranosyluronate)-a-D-glucopyranoside (Vb)
E F To a solution of compound IVb (385 mg, 0.56 mmol, 1 .0 eq.) in chloroform (stabilised with amylene, 30 mL) were added, under an argon atmosphere, P205 (410 mg, 2.80 mmol, 5.0 eq.) and propargylaldehyde diethylacetal (0.4 mL, 2.79 mmol, 5.0 eq.), then the reaction mixture was heated at reflux. After 5 hours stirring, the reaction mixture was filtered through a pad of Celite® then the solvent was removed under vacuum. The crude mixture was suspended in EtOAc (60 mL), washed with a 5% NaHC03 aqueous solution (1 x 15 mL) and brine (1 x 15 mL). The organic layer was dried (MgS04), filtered, and evaporated. Column chromatography (gradient hexane/ethyl acetate 90:10 – 70:30) afforded compound Vb as a colourless oil (275 mg, 0.36 mmol, 64%) in a diastereomeric mixture (64:36).
1H NMR (CDCI3, 250 MHz): δ 1.17-1 .27 (m, 3H), 2.55 (d, 0.36H), 2.57 (d, 0.64H), 2.92- 3.08 (m, 2H), 3.38 (s, 3H), 3.49 (s, 1.92H), 3.50 (s, 1.08H), 3.57 (s, 1.08H), 3.59 (s, 1.92H), 3.60 (s, 1.92H), 3.62 (s, 1.08H), 3.44-3.97 (m, 10H), 4.35 (t, 1 H), 4.46-4.76 (m, 6H), 5.03 (d, 1 H), 5.32 (d, 0.36H), 5.56 (d, 0.64H), 7.21-7.42 (m, 15H).
Elemental analysis: Calculated: C: 65.95 ; H: 6.85. Found: C: 65.92 ; H: 6.75.
ESI-MS (pos. mode): m/z = 787 [M+Na]+.
Preparation of Methyl-2,3,6-tri-0-benzyl-4-0(4′-0-(1 “-ethoxy-2″-propyn-1 ” 1 ‘,2′,3′-tri-0-methyl-a-D-glucopyranosiduronic acid)-a-D-glucopyranoside (Vlb)
E F
To a solution of compound Vb (1.02 g, 1.33 mmol, 1.0 eq.) in EtOH/H20 (1 :1 , 100 mL) was added sodium hydroxide (82 mg, 2.05 mmol, 1.5 eq.). After 3 hours stirring at room temperature sodium hydroxide was added anew (27 mg, 0.68 mmol, 0.5 eq.). After an additional hour stirring the solvent was evaporated. The residue obtained was dissolved in water (40 mL). The pH of the aqueous layer was reduced to 2-3 with a 10% HCI aqueous solution then the layer was saturated with sodium chloride before extraction with dichloromethane (3 x 20 mL). The combined organics were dried (MgS04), filtered and removed under vacuum. Compound VIb was obtained without further purification as a white solid (930 mg, 1.24 mmol, 93%), in a diastereomeric mixture (63:37).
1H NMR (CDCIs, 400 MHz): δ 1.17-1.28 (m, 3H), 2.65 (d, 0.63H), 2.68 (d, 0.37H), 2.90- 3.09 (m, 2H), 3.37 (s, 3H), 3.46 (s, 1.1 1 H), 3.57 (s, 1.89H), 3.58 (s, 1.1 1 H), 3.60 (s, 1.89H), 3.42-3.90 (m, 10H), 4.29 (d, 1 H), 4.46-4.97 (m, 7H), 5.44 (d, 0.37H), 5.61 (d, 0.63H), 7.28-7.44 (m, 15H).
ESI-MS (pos. mode): m/z = 773 [M+Na]+ , 795 [M-H+2Na]+ . ESI-MS (neg. mode): m/z = 749 [M-H]\
Preparation of Methyl-2,3,6-tri-0-benzyl-4-0(4′,7′-anhydro-6′-deoxy-6′-methylene-7′- ethoxy-2 3′-di-0-methyl-α-L-/‘ o-heptopyranosyl)-α-D-glucopyranoside (Vllb)
To a solution of compound VIb (647 mg, 0.86 mmol, 1.0 eq.) in anhydrous THF (20 mL) and cooled to 0°C, were added IBCF (0.1 1 mL, 0.85 mmol, 1.0 eq.) and N- methylmorpholine (0.10 mL, 0.91 mmol, 1.1 eq.). After 10 minutes stirring, the flask was covered with aluminium foil, 2-mercaptopyridine /V-oxide sodium salt (512 mg, 3.43 mmol, 4.0 eq.) was added and the reaction mixture was stirred at ambient temperature. After 20 minutes anhydrous THF (100 mL) then ie f-butylthiol (0.18 mL, 1 .68 mmol, 2.0 eq.) were added. The aluminium foil was removed and the reaction mixture was irradiated and heated 15 minutes with a UV lamp (300W). The thiol excess was neutralized with a NaOCI aqueous solution (13%, 10 mL). The reaction mixture was concentrated then dissolved in EtOAc (100 mL), washed successively with a 5% NaHC03 aqueous solution (1 x 15 mL), a 5 % citric acid aqueous solution (1 x 15 mL) and brine (1 x 15 mL), then the aqueous layer was extracted with dichloromethane (2 x 20 mL). The combined organics were dried (MgS04), filtered and concentrated. Column chromatography (gradient hexane/ethyl acetate 90 :10 – 70:30) afforded compound Vllb as a colourless oil (251 mg, 0.36 mmol, 42%) in a mixture of diastereomers (61 :39).
1H NMR (CDCI3, 250 MHz): δ 1.18-1.29 (m, 3H), 2.90-3.07 (m, 1 H), 3.37 (s, 1.17H), 3.38 (s, 1 .83H), 3.46 (s, 3H), 3.54 (s, 1.83H), 3.60 (s, 1 .17H), 3.29-4.00 (m, 10H), 4.1 1 -4.26 (m, 1 H), 4.50-4.96 (m, 8H), 5.09-5.48 (m, 3H), 7.23-7.39 (m, 15H).
ESI-MS (pos. mode): m/z = 720 [M+Na]+ .
Preparation of Methyl-2,3,6-tri-0-benzyl-4-0(4′,7′-anhydro-7′-ethoxy-2′,3′-di-0- methyl-a-L-/‘ o-heptopyranosid-6′-ulosyl)-a-D-glucopyranoside (Vlllb)
Through a solution of compound Vllb (145 mg, 0.21 mmol, 1 .0 eq.) in anhydrous dichloromethane (10 mL), under an argon atmosphere and cooled to -78°C, was bubbled ozone (0.2 L/min, 1 10 V). When the solution had turned dark blue, oxygen was bubbled through in order to remove the excess ozone. When the solution became colorless dimethylsulfide (4 drops) was added and the solution was brought to room temperature. After 30 min stirring the reaction mixture was concentrated. Column chromatography (gradient hexane/ethyl acetate 90:10 – 60:40) afforded compound Vlllb as a white solid (100 mg, <67%) in a mixture of diastereomers (67:33).
1H NMR (CDCI3, 400 MHz): δ 1.21 -1 .28 (m, 3H), 2.93-3.07 (m, 1 H), 3.31-4.26 (m, 20H), 4.52-5.02 (m, 9H), 7.20-7.45 (m, 15H).
ESI-MS (pos. mode): m/z = 731 [M+Na]+ .
Preparation of Methyl-2,3,6-tri-0-benzyl-4-0(methyl 2′,3′-di-0-methyl-a-L- idopyranosiduronate)-a-D-glucopyranoside (IXb)
To a solution of compound Vlllb (62 mg, 87 μηηοΙ, 1 .0 eq.) in dichloromethane (5 mL), under an argon atmosphere and cooled to 0°C, were added m-CPBA (77%, 58 mg, 259 μηηοΙ, 3.0 eq.) and NaHC03 (1 1 mg, 130 μηηοΙ, 1 .5 eq.). After 5 hours stirring the solvent was removed under vacuum. The reaction mixture was then dissolved in EtOAc (50 mL) and washed successively with a 5% NaHC03 aqueous solution (1 x 10 mL), a 5 % citric acid aqueous solution (1 x 10 mL) and brine (1 x 10 mL). The organic layer was dried (MgS04), filtered and concentrated. The crude mixture was dissolved in anhydrous methanol (10 mL) and sodium methoxide was added to reach pH = 10. After 30 minutes stirring at room temperature the reaction mixture was neutralized with Dowex®, filtered through a pad of Celite®, and concentrated. The residue obtained was dissolved in DMF (10 mL) then triethylamine (13 μί, 93 μηηοΙ, 1.1 eq.) and methyl iodide (27 μί, 434 μηηοΙ, 5.0 eq.) were added. After 2h30 stirring the reaction mixture was concentrated, dissolved in EtOAc (40 mL) and washed with a 5% citric acid aqueous solution (2 x 10 mL), a 5% NaHC03 aqueous solution (2 x 10 mL), and brine (1 x 10 mL). The organic layer was dried (MgS04), filtered and concentrated. Column chromatography (gradient hexane/ethyl acetate 60:40-50:50) afforded compound IXb as a colourless oil (12 mg, 18 μηηοΙ, 20% over two steps).
1H NMR (CDCIs, 400 MHz): δ 3.23 (s, 3H), 3.20-3.25 (m, 1 H), 3.36 (s, 3H), 3.43 (s, 3H), 3.46 (s, 3H), 3.33-3.58 (m, 3H), 3.60-3.69 (m, 2H), 3.72-3.95 (m, 4H), 4.53-4.60 (m, 4H), 4.68-4.97 (m, 4H), 5.14 (s, 1 H), 7.24-7.37 (15H).
ESI-MS (pos. mode): m/z = 705 [M+Na]+ .
……………
Volume 69, Issue 15, 15 April 2013, Pages 3149–3158
Abstract
Idraparinux, the fully O-sulfated, O-methylated, heparin-related pentasaccharide possessing selective factor Xa inhibitory activity, was prepared by two novel synthetic pathways. Each route was based on a 2+3 block synthesis utilizing the same l-iduronic acid-containing trisaccharide acceptor, which was glycosylated with either a glucuronide disaccharide donor or its non-oxidized precursor. The latter route, involving the oxidation of the glucose unit into d-glucuronic acid at a pentasaccharide level proved to be much more efficient, providing the target pentasaccharide in a reasonable overall yield.
Graphical abstract

- ……………………………
- SYNTHESIS
- US 20120041189 A1,
- http://www.patexia.com/us-publications/20120041189
- EXAMPLE 1Preparation of the Compound of Formula (I) in Crystalline Form (Scheme 1)1.1: Preparation of the Compound of Formula (I′)The compound of formula (I″) is obtained, for example, according to the teaching of patent EP 0 529 715 B1 or of the articles “Bioorg. Med. Chem.” (1994, Vol. 2, No. 11, pp. 1267-1280), “Bioorg. Med. Chem. Letters” (1992, Vol. 2, No. 9, pp. 905-910) or “Magnetic Resonance in Chemistry” (2001, Vol. 39, pp. 288-293). The compound of formula (I″) (5 g, 3.06 mmol) is dissolved in acetonitrile (10 mL). Deionized water (12.2 mL) and aqueous 30% sodium hydroxide solution (4.1 g) are then added. The mixture is heated to 40° C. and maintained at this temperature for 5 hours. The reaction medium is then cooled to 20° C. and acidified to pH 6.25 with aqueous 1N hydrochloric acid solution (about 17.7 g) before extraction with MTBE of certain impurities, the saponified product remaining in the aqueous phase. The residual acetonitrile, contained in the aqueous phase, is then removed by concentration, followed by diluting with deionized water (125 mL). The saponified product is finally precipitated at pH 1.5 by adding aqueous 1N hydrochloric acid solution (about 17.6 g) at 20° C. The suspension is maintained for 4 hours at 20° C. before filtration. The wet solid is finally dried in a vacuum oven at 30° C. to give 2.93 g (93.6%) of compound of formula (I).NMR (anomeric protons of the saccharide units D, E, F, G, H): 5.79, 5.14, 5.55, 5.92, 4.94 ppm.1.2 Preparation of the Crude Compound of Formula (I)The compound of formula (I′) obtained after the preceding step is dissolved in tetrahydrofuran (18 mL). Palladium-on-charcoal (0.3 g) is added. The reaction medium is hydrogenated at 0.3 bar of hydrogen (relative pressure) for 4 hours. After filtering and evaporating, 2.12 g (99%) of the crude compound of formula (I) are obtained.1.3: Preparation of the Compound of Formula (I) in Crystalline Form Using an Isopropanol/MTBE MixtureThe crude hydrogenated product obtained after the preceding step is dissolved in isopropanol (13 mL) at 65° C., and then crystallized at room temperature. The suspension is then cooled to 40° C., followed by addition of MTBE (13 mL), and is then cooled slowly to 10° C. After maintenance at 10° C. for 2 hours, the crystalline hydrogenated product is filtered off, washed and dried. 1.66 g of the compound of formula (I) in crystalline form are thus obtained, in the form of a cream-white powder. The reaction yield for the production of the compound of formula (I) in crystalline form, from the compound of formula (I′), is 92.5%. When expressed relative to the starting compound (I″), the reaction yield for the production of the compound of formula (I) in crystalline form is 86.6%.NMR (anomeric protons of the saccharide units D, E, F, G, H) of the compound of formula (I) in crystalline form: 5.77, 5.11, 5.51, 5.84, 5.01 ppm.1.4: Preparation of the Compound of Formula (I) in Crystalline Form Using IsopropanolThe crude hydrogenated product obtained after step 1.2 is dissolved in isopropanol (5 volumes) at 75° C. The medium is then cooled slowly until crystals appear, according to the known standard techniques for crystallization. The process is performed, for example, by a first step of cooling at 65° C. for 1 hour, and than a second step of cooling to a final temperature of 25° C. over 4 hours or of 5° C. over 6 hours, and finally maintenance at this final temperature for 30 minutes. The suspension is then filtered and rinsed with isopropanol (2×0.1 V) and compound (I) is isolated in the form of white crystals, which appear under a microscope in the form of needles. The 1H NMR analysis of these crystals is identical to that described after step 1.3 above.EXAMPLE 4Preparation of Idraparinux from the Compound of Formula (I) in Crystalline Form (Scheme 2)The preparation of idraparinux (II) from the compound of formula (I) is summarized in Scheme 2.The compound of formula (I) in crystalline form, as obtained according to Example 1.3, is dissolved in N,N’-dimethylformamide (6.6 mL) and then heated to 30.degree. C. Under an inert atmosphere, 3.8 g of pyridine-sulfur trioxide complex are added slowly, followed by maintenance at 30.degree. C. for 4 hours. The reaction medium is then poured into aqueous 23.8% sodium hydrogen carbonate solution (16.3 g) maintained at a maximum of 25.degree. C., to obtain the compound of formula (II). The reaction medium is kept stirring for hours. The solution of sulfated product is then poured onto an MTBE/isopropanol/ethanol mixture (171 mL/70 mL/70 mL). Precipitation of the product is observed, and, after filtering off, washing and drying the cake, 4.99 g (96.8%) of compound of formula (II) are obtained, and are then purified by anion-exchange chromatography according to the usual techniques.
NMR (anomeric protons of the saccharide units D, E, F, G, H) of the compound of formula (II): 5.48, 4.68, 5.44, 5.08, 5.18 ppm.It thus appears that the process according to the invention makes it possible to obtain idraparinux (compound of formula (II)) in a chemical yield of about 84% (precisely 83.8% according to the protocols described above) starting from the compound of formula (I”), i.e. a gain in yield of about 30% relative to the process described in patent EP 0 529 715 B1.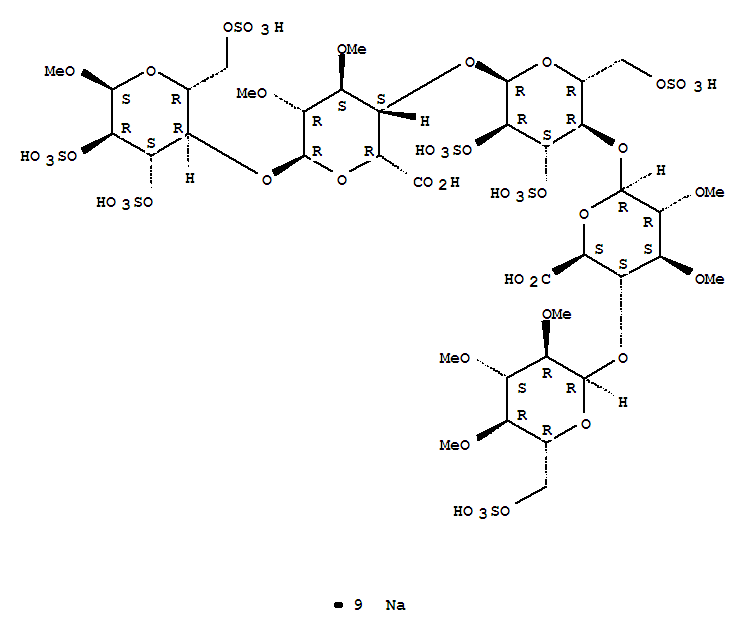 IDRAPARINUX
IDRAPARINUX
References
- Bousser MG, Bouthier J, Büller HR, et al. (January 2008). “Comparison of idraparinux with vitamin K antagonists for prevention of thromboembolism in patients with atrial fibrillation: a randomised, open-label, non-inferiority trial”. Lancet 371 (9609): 315–21. doi:10.1016/S0140-6736(08)60168-3.PMID 18294998.
- Buller HR, Cohen AT, Davidson B, et al. (September 2007). “Idraparinux versus standard therapy for venous thromboembolic disease”. N. Engl. J. Med. 357 (11): 1094–104. doi:10.1056/NEJMoa064247. PMID 17855670.
- Bioorg Med Chem1994,2,(11):1267
- Drugs Fut2002,27,(7):639
- EP 0454220, JP 1992225994, US 5378829, US 5382570, US 5529985, US 5773605
- EP 0529715
- Bioorg Med Chem Lett. 2009 Jul 15;19(14):3875-9. doi: 10.1016/j.bmcl.2009.03.155. Epub 2009 Apr 5.
- Chemistry – A European Journal, 2012 , vol. 18, 34 p. 10643 – 10652
- Magnetic Resonance in Chemistry, 2001 , vol. 39, 5 p. 288 – 293
- Tetrahedron, 2013 , vol. 69, 15 p. 3149 – 3158…….. MP 210-15 DEG CENT
- I. Capila, R.J. Linhardt Angew. Chem., Int. Ed., 41 (2002), p. 390
- I. Capila, R.J. Linhardt
- L. Roden D.A. Lane, U. Lindahl (Eds.), Chemical and Biological Properties, Clinical Applications, CRC Press, Boca Raton, FL (1989), p. 1
- L. Roden
- (a) C.A.A. van Boeckel, M. Petitou Angew. Chem., Int. Ed. Engl., 32 (1993), p. 1671
- (b) M. Petitou, C.A.A. van Boeckel Angew. Chem., Int. Ed., 43 (2004), p. 3118
- (a) C.A.A. van Boeckel, M. Petitou
- M. Petitou, B. Casu, U. Lindahl Biochimie, 85 (2003), p. 83Article |
- M. Petitou, B. Casu, U. Lindahl
- (a) M. Petitou, P. Duchaussoy, I. Lederman, J. Choay, J.C. Jacquinet, P. Sinay, G. Torri Carbohydr. Res., 167 (1987), p. 67Article |
- (b) P.-A. Driguez, I. Lederman, J.-M. Strassel, J.-M. Herbert, M. Petitou J. Org. Chem., 64 (1999), p. 9512
- (c) J. Choay, M. Petitou, J.C. Lormeau, P. Sinay, B. Casu, G. GattiBiochem. Biophys. Res. Commun., 116 (1983), p. 492Article |
- (d) P. Sinay, J.C. Jacquinet, M. Petitou, P. Duchaussoy, I. Lederman, J. Choay, G. Torri Carbohydr. Res., 132 (1984), p. C5
- (a) M. Petitou, P. Duchaussoy, I. Lederman, J. Choay, J.C. Jacquinet, P. Sinay, G. Torri
- (a) P. Westerduin, C.A.A. van Boeckel, J.E.M. Basten, M.A. Broekhoven, H. Lucas, A. Rood, H. van der Heijden, R.G.M. van Amsterdam, T.G. van Dinther, D.G. Meuleman, A. Visser, G.M.T. Vogel, J.B.L. Damm, G.T. Overklift
- Bioorg. Med. Chem., 2 (1994), p. 1267Article |
 PDF (1579 K)
PDF (1579 K)
- (b) J.M. Herbert, J.P. Herault, A. Bernat, R.G.M. van Amsterdam, J.C. Lormeau, M. Petitou, C. van Boeckel, P. Hoffmann, D.G. Meuleman Blood, 91 (1998), p. 4197
- (a) P. Prandoni, D. Tormene, M. Perlati, B. Brandolin, L. SpieziaExpert Opin. Investig. Drugs, 17 (2008), p. 773
- (b) A.S. Go, D.E. Singer Lancet, 371 (2008), p. 278Article|
- (a) P. Prandoni, D. Tormene, M. Perlati, B. Brandolin, L. Spiezia
- I.M. Pinilla, M.B. Martinez, J.A. Galbis Carbohydr. Res., 338 (2003), p. 549Article |
- I.M. Pinilla, M.B. Martinez, J.A. Galbis
- (a) K. Yoza, N. Amanokura, Y. Ono, T. Akao, H. Shinmori, M. Takeuchi, S. Shinkai, D.N. Reinhoudt Chem. Eur. J., 5 (1999), p. 2722
- (b) J. Elhalabi, K.G. RiceCarbohydr. Res., 335 (2001), p. 159Article|
 PDF (177 K)
PDF (177 K)
- (a) K. Yoza, N. Amanokura, Y. Ono, T. Akao, H. Shinmori, M. Takeuchi, S. Shinkai, D.N. Reinhoudt
- S.D. Debenham, E.J. Toone Tetrahedron: Asymmetry, 11 (2000), p. 385Article |
- S.D. Debenham, E.J. Toone
- A. Meijer, U. Ellervik J. Org. Chem., 69 (2004), p. 6249 and references therein
- A. Meijer, U. Ellervik
- P. Duchaussoy, G. Jaurand, P.-A. Driguez, I. Lederman, F. Gourvenec, J.-M. Strassel, P. Sizun, M. Petitou, J.-M. HerbertCarbohydr. Res., 317 (1999), p. 63Article |
- P. Duchaussoy, G. Jaurand, P.-A. Driguez, I. Lederman, F. Gourvenec, J.-M. Strassel, P. Sizun, M. Petitou, J.-M. Herbert
- J. Lee, X.-A. Lu, S.S. Kulkarni, Y. Wen, S.-C. Hung J. Am. Chem. Soc., 126 (2004), p. 476
- J. Lee, X.-A. Lu, S.S. Kulkarni, Y. Wen, S.-C. Hung
MORE
- L.A.G.M. van den Broek, D.J. Vermaas, B.M. Heskamp, C.A.A. van Boeckel Recl. Trav. Chim. Pays-Bas., 112 (1993), p. 82
- R.R. Schmidt, J. Michel Angew. Chem., Int. Ed. Engl., 19 (1980), p. 731
- L.A.G.M. van den Broek, D.J. Vermaas, B.M. Heskamp, C.A.A. van Boeckel
- (a) F. Lin, W. Peng, W. Xu, X. Han, B. Yu Carbohydr. Res., 339 (2004), p. 1219
- Article |
 PDF (270 K)
PDF (270 K)
- (b) P.L. Anelli, C. Biffi, F. Montanari, S. Quici J. Org. Chem., 52 (1987), p. 2559
- (c) P.L. Anelli, S. Banfi, F. Montanari, S. Quici J. Org. Chem., 54 (1989), p. 2970
- (a) F. Lin, W. Peng, W. Xu, X. Han, B. Yu
- (a) P. Bourhis, F. Machetto, P. Duchaussoy, J.-P. Herault, J.-M. Mallet, J.-M. Herbert, M. Petitou, P. Sinay
- For other examples of 4,6-locked idose glycosyl donors, see: Bioorg. Med. Chem. Lett., 7 (1997), p. 2843
- Article |
 PDF (244 K)
PDF (244 K)
- (b) N. Barroca, J.-C. Jacquinet Carbohydr. Res., 337 (2002), p. 673
- Article |
 PDF (300 K)
PDF (300 K)
- (c) J. Kuszmann, G. Medgyes, S. Boros Carbohydr. Res., 339 (2004), p. 1569
- Article |
 PDF (352 K)
PDF (352 K)
- (d) J. Tatai, P. Fugedi Org. Lett., 9 (2007), p. 4647
| WO2002024754A1 | Sep 20, 2001 | Mar 28, 2002 | Akzo Nobel Nv | Polysaccharides with antithrombotic activity comprising at least a covalent bond with biotin or a biotin derivative |
| WO2006030104A1 * | Sep 7, 2005 | Mar 23, 2006 | Sanofi Aventis | Biotinylated hexadecasaccharides, preparation and use thereof |
| WO2007042469A2 * | Oct 6, 2006 | Apr 19, 2007 | Organon Nv | Anticoagulant antithrombotic dual inhibitors comprising a biotin label |
| EP0230023A2 * | Dec 19, 1986 | Jul 29, 1987 | Marion Merrell Dow Inc. | Pharmaceutical compositions for the enhancement of wound healing |
| EP0300099A1 * | Jul 20, 1987 | Jan 25, 1989 | Akzo N.V. | New pentasaccharides |
| EP0301618A2 * | Jul 4, 1988 | Feb 1, 1989 | Akzo N.V. | New pentasaccharides |
| EP0454220A1 * | Apr 16, 1991 | Oct 30, 1991 | Akzo Nobel N.V. | Carbohydrate derivatives comprising a trisaccharide unit |
| GB1110939A * | Title not available | |||
| US3017407 * | Aug 18, 1958 | Jan 16, 1962 | Riker Laboratories Inc | Process for producing polysulfuric acid esters of polysaccharides |


2. FONDAPARINUX
FONDAPARINUX

FONDAPARINUX
Fondaparinux is a drug belonging to the group of the antithrombotic agents and are used to prevent deep vein thrombosis in patients undergoing orthopedic surgery. It is also used for the treatment of severe venous thrombosis and pulmonary
114870-03-0 ………..10x SODIUM SALT
| CAS NUMBER | 114870-03-0 FREE FORM |
|---|
| MF | C31H43N3Na10O49S8 10X SODIUM |
|---|---|
| MW | 1726.77 g/mol 10X SODIUM |
GSK-576428 Org-31540 SR-90107SR-90107A
launched 2002
Arixtra, Quixidar, Fondaparinux sodium, Fondaparin sodium, Arixtra (TN), Fondaparinux, Org-31540, AC1LCS4W, SR-90107A
Fondaparinux (Arixtra) is a synthetic pentasaccharide anticoagulant. Apart from the O-methyl group at the reducing end of the molecule, the identity and sequence of the five monomeric sugar units contained in fondaparinux is identical to a sequence of five monomeric sugar units that can be isolated after either chemical or enzymatic cleavage of the polymeric glycosaminoglycan heparin and heparan sulfate (HS). This monomeric sequence in heparin and HS is thought to form the high affinity binding site for the natural anti-coagulant factor, antithrombin III (ATIII).
Binding of heparin/HS to ATIII has been shown to increase the anti-coagulant activity of antithrombin III 1000-fold. Fondaparinux potentiates the neutralizing action ofATIII on activated Factor X 300-fold. Fondaparinux may be used: to prevent venous thromboembolism in patients who have undergone orthopedic surgery of the lower limbs (e.g. hip fracture, hip replacement and knee surgery); to prevent VTE in patients undergoing abdominal surgery who are are at high risk of thromboembolic complications; in the treatment of deep vein thrombosis (DVT) and pumonary embolism (PE); in the management of unstable angina (UA) and non-ST segment elevation myocardial infarction (NSTEMI); and in the management of ST segment elevation myocardial infarction (STEMI).

FONDAPARINUX
Fondaparinux (trade name Arixtra) is an anticoagulant medication chemically related to low molecular weight heparins. It is marketed byGlaxoSmithKline. A generic version developed by Alchemia is marketed within the US by Dr. Reddy’s Laboratories.
Fondaparinux is a synthetic pentasaccharide Factor Xa inhibitor. Apart from the O-methyl group at the reducing end of the molecule, the identity and sequence of the five monomeric sugar units contained in fondaparinux is identical to a sequence of five monomeric sugar units that can be isolated after either chemical or enzymatic cleavage of the polymeric glycosaminoglycans heparin and heparan sulfate (HS). Within heparin and heparan sulfate this monomeric sequence is thought to form the high affinity binding site for the anti-coagulant factor antithrombin III (ATIII). Binding of heparin/HS to ATIII has been shown to increase the anti-coagulant activity of antithrombin III 1000 fold. In contrast to heparin, fondaparinux does not inhibit thrombin.
Fondaparinux is given subcutaneously daily. Clinically, it is used for the prevention of deep vein thrombosis in patients who have had orthopedic surgery as well as for the treatment of deep vein thrombosis and pulmonary embolism.
One potential advantage of fondaparinux over LMWH or unfractionated heparin is that the risk for heparin-induced thrombocytopenia (HIT) is substantially lower. Furthermore, there have been case reports of fondaparinux being used to anticoagulate patients with established HIT as it has no affinity to PF-4. However, its renal excretion precludes its use in patients with renal dysfunction.
Unlike direct factor Xa inhibitors, it mediates its effects indirectly through antithrombin III, but unlike heparin, it is selective for factor Xa.[1]
Fondaparinux is similar to enoxaparin in reducing the risk of ischemic events at nine days, but it substantially reduces major bleeding and improves long term mortality and morbidity.[2]
It has been investigated for use in conjunction with streptokinase.[3]
Fondaparinux sodium, a selective coagulation factor Xa inhibitor, was first launched in the U.S. in 2002 by GlaxoSmithKline in a subcutaneous injection formulation for the prophylaxis of deep venous thrombosis (DVT) which may lead to pulmonary embolism in patients at risk for thromboembolic complications who are undergoing hip replacement, knee replacement, hip fracture surgery or abdominal surgery. The product is available in Japan for the treatment of acute deep venous thrombosis and acute pulmonary thromboembolism. In 2004, GlaxoSmithKline launched fondaparinux as an injection to be used in conjunction with warfarin sodium for the subcutaneous treatment of acute pulmonary embolism and DVT.
In 2007, GlaxoSmithKline received approval in the E.U. for the treatment of acute coronary syndrome (ACS), specifically unstable angina or non-ST segment elevation myocardial infarction (UA/NSTEMI) and ST-segment elevation myocardial infarction (STEMI), while in the U.S. an approvable letter was received for this indication. Currently, the drug is in clinical development at GlaxoSmithKline for the treatment of venous limb superficial thrombosis.
GlaxoSmithKline had filed a regulatory application in the E.U. seeking approval of fondaparinux sodium for the prevention of venous thromboembolic events (VTE), however; in 2008, the application was withdrawn for commercial reasons. Commercial launch in Japan for the product for the prevention of venous thromboembolism in high risk patients undergoing surgery in the abdomen took place in 2008.
In 2010, the EMA approved a regulatory application filed by GlaxoSmithKline seeking approval of a once-daily formulation of fondaparinux sodium for the treatment of adults with acute symptomatic spontaneous superficial-vein thrombosis (SVT) of the lower limbs without concomitant DVT. Product launch took place in the U.K. for this indication the same year.
The antithrombotic activity of fondaparinux is the result of antithrombin III (ATIII)-mediated selective inhibition of Factor Xa. By selectively binding to ATIII, the drug potentiates (about 300 times) the innate neutralization of Factor Xa by ATIII. Neutralization of Factor Xa, in turn, interrupts the blood coagulation cascade and thus inhibits thrombin formation and thrombus development. Fondaparinux does not inactivate thrombin (activated Factor II) and has no known effect on platelet function. At the recommended dose, no effects have been demonstrated on fibrinolytic activity or bleeding time.
Originally developed by Organon and Sanofi (formerly known as sanofi-aventis), fondaparinux sodium is currently available in approximately 30 countries. In 2004, Organon transferred its rights to the drug to Sanofi in exchange for revenues based on future sales from jointly developed antithrombotic products and in early 2005, GlaxoSmithKline also acquired the antithrombotic.
At the beginning of 2005, GlaxoSmithKline signed a two-year agreement with Adolor (acquired by Cubist in 2011) for the copromotion of fondaparinux sodium in the U.S. In Sepetember 2013, Aspen Pharmacare acquired Arixtra global rights (excluding China, India and Pakistan) from GlaxoSmithKline for the treatment of thrombosis with GlaxoSmithKline commercializing the product in Indonesia under licence from Aspen.
Chemical structure

Abbreviations
- GlcNS6S = 2-deoxy-6-O-sulfo-2-(sulfoamino)-α-D-glucopyranoside
- GlcA = β-D-glucopyranuronoside
- GlcNS3,6S = 2-deoxy-3,6-di-O-sulfo-2-(sulfoamino)-α-D-glucopyranosyl
- IdoA2S = 2-O-sulfo-α-L-idopyranuronoside
- GlcNS6SOMe = methyl-O-2-deoxy-6-O-sulfo-2-(sulfoamino)-α-D-glucopyranoside
The sequence of monosaccharides is D-GlcNS6S-α-(1,4)-D-GlcA-β-(1,4)-D-GlcNS3,6S-α-(1,4)-L-IdoA2S-α-(1,4)-D-GlcNS6S-OMe, as shown in the following structure:
ARIXTRA (fondaparinux sodium) Injection is a sterile solution containing fondaparinux sodium. It is a synthetic and specific inhibitor of activatedFactor X (Xa). Fondaparinux sodium is methyl O-2-deoxy-6-O-sulfo-2-(sulfoamino)-α-D-glucopyranosyl-(1→4)-O-β-D-glucopyranuronosyl-( 1→4)-O-2-deoxy-3,6-di-O-sulfo-2-(sulfoamino)-α-D-glucopyranosyl-(1→4)-O-2-Osulfo-α-L-idopyranuronosyl-(1→4)-2-deoxy-6-O-sulfo-2-(sulfoamino)-α-D-glucopyranoside, decasodium salt.
The molecular formula of fondaparinux sodium is C31H43N3Na10O49S8 and its molecular weight is 1728. The structural formula is provided below:

ARIXTRA is supplied as a sterile, preservative-free injectable solution for subcutaneous use.
Each single-dose, prefilled syringe of ARIXTRA, affixed with an automatic needle protection system, contains 2.5 mg of fondaparinux sodium in 0.5 mL, 5.0 mg of fondaparinux sodium in 0.4 mL, 7.5 mg of fondaparinux sodium in 0.6 mL, or 10.0 mg of fondaparinux sodium in 0.8 mL of an isotonic solutionof sodium chloride and water for injection. The final drug product is a clear and colorless to slightly yellow liquid with a pH between 5.0 and 8.0.
Molecular formula of fondaparinux sodium is C31H43N3Na10O49S8
Chemical IUPAC Name is decasodium (2R,3S,4S,5R,6R)-3-[(2R,3R,4R,5S,6R)-5-[(2R,3R,4S,5S,6S)-6- carboxylato-5-[(2R,3R,4R,5S,6R)- 4,5-dihydroxy-3- (sulfonatoamino)-6-(sulfonatooxymethyl)oxan-2-yl]oxy-3,4- dihydroxy-oxan-2-yl]oxy-3-(sulfonatoamino)-4- sulfonatooxy-6-(sulfonatooxymethyl)oxan-2-yl]oxy- 4-hydroxy-6-[(2R,3S,4R,5R,6S)-4-hydroxy-6- methoxy-5-(sulfonatoamino)-2-(sulfonatooxymethyl) oxan-3-yl]oxy-5-sulfonatooxy-oxane-2-carboxylate
Molecular weight is 1726.77 g/mol
……………….
INTRODUCTION
In U.S. Patent No. 7,468,358, Fondaparinux sodium is described as the “only anticoagulant thought to be completely free of risk from HIT-2 induction.” The biochemical and pharmacologic rationale for the development of a heparin pentasaccharide in Thromb. Res., 86(1), 1-36, 1997 by Walenga et al. cited the recently approved synthetic pentasaccharide Factor Xa inhibitor Fondaparinux sodium. Fondaparinux has also been described in Walenga et al., Expert Opin. Investig. Drugs, Vol. 11, 397-407, 2002 and Bauer, Best Practice & Research Clinical Hematology, Vol. 17, No. 1, 89-104, 2004.
Fondaparinux sodium is a linear octasulfated pentasaccharide (oligosaccharide with five monosaccharide units ) molecule having five sulfate esters on oxygen (O-sulfated moieties) and three sulfates on a nitrogen (N- sulfated moieties). In addition, Fondaparinux contains five hydroxyl groups in the molecule that are not sulfated and two sodium carboxylates. Out of five saccharides, there are three glucosamine derivatives and one glucuronic and one L-iduronic acid. The five saccharides are connected to each other in alternate α and β glycosylated linkages (see Figure 1).
Figure 1 Fondaparinux Sodium
Monosaccharide E Monosaccharide D Monosaccharide C Monosaccharide B Monosaccharide A derived from derived from derived from derived from derived from
Monomer E Monomer D Monomer C Monomer B1 Monomer A2
Fondaparinux Sodium
Fondaparinux sodium is a chemically synthesized methoxy derivative of the natural pentasaccharide sequence, which is the active site of heparin that mediates the interaction with antithrombin (Casu et al., J. Biochem., 197, 59, 1981). It has a challenging pattern of O- and N- sulfates, specific glycosidic stereochemistry, and repeating units of glucosamines and uronic acids (Petitou et al, Progress in the Chemistry of Organic Natural Product, 60, 144-209, 1992).
The monosaccharide units comprising the Fondaparinux molecule are labeled as per the convention in Figure 1, with the glucosamine unit on the right referred to as monosaccharide A and the next, an uronic acid unit to its left as B and subsequent units, C, D and E respectively. The chemical synthesis of Fondaparinux starts with monosaccharides of defined structures that are themselves referred to as Monomers A2, Bl, C, D and E, for differentiation and convenience, and they become the corresponding monosaccharides in fondaparinux sodium.
Due to this complex mixture of free and sulfated hydroxyl groups, and the presence of N- sulfated moieties, the design of a synthetic route to Fondaparinux requires a careful strategy of protection and de-protection of reactive functional groups during synthesis of the molecule. Previously described syntheses of Fondaparinux all adopted a similar strategy to complete the synthesis of this molecule. This strategy can be envisioned as having four stages.
The strategy in the first stage requires selective de-protection of five out of ten hydroxyl groups. During the second stage these five hydroxyls are selectively sulfonated. The third stage of the process involves the de -protection of the remaining five hydroxyl groups. The fourth stage of the process is the selective sulfonation of the 3 amino groups, in the presence of five hydroxyl groups that are not sulfated in the final molecule. This strategy can be envisioned from the following fully protected pentasaccharide, also referred to as the late-stage intermediate.
In this strategy, all of the hydroxyl groups that are to be sulfated are protected with an acyl protective group, for example, as acetates (R = CH3) or benzoates (R = aryl) (Stages 1 and 2) All of the hydroxyl groups that are to remain as such are protected with benzyl group as benzyl ethers (Stage 3). The amino group, which is subsequently sulfonated, is masked as an azide (N3) moiety (Stage 4). R1 and R2 are typically sodium in the active pharmaceutical compound (e.g., Fondaparinux sodium).
This strategy allows the final product to be prepared by following the synthetic operations as outlined below: a) Treatment of the late- stage intermediate with base to hydrolyze (deprotect) the acyl ester groups to reveal the five hydroxyl groups. The two R1 and R2 ester groups are hydrolyzed in this step as well.
b) Sulfonation of the newly revealed hydroxyl groups.
c) Hydrogenation of the O-sulfated pentasaccharide to de-benzylate the five benzyl- protected hydroxyls, and at the same time, unmask the three azides to the corresponding amino groups.
d) On the last step of the operation, the amino groups are sulfated selectively at a high pH, in the presence of the five free hydroxyls to give Fondaparinux (Figure 1). While the above strategy has been shown to be viable, it is not without major drawbacks. One drawback lies in the procedure leading to the fully protected pentasaccharide (late stage intermediate), especially during the coupling of the D-glucuronic acid to the next adjacent glucose ring (the D-monomer to C-monomer in the EDCBA nomenclature shown in Figure 1). Sugar oligomers or oligosaccharides, such as Fondaparinux, are assembled using coupling reactions, also known as glycosylation reactions, to “link” sugar monomers together. The difficulty of this linking step arises because of the required stereochemical relationship between the D-sugar and the C-sugar, as shown below:
The stereochemical arrangement illustrated above in Figure 2 is described as having a β- configuration at the anomeric carbon of the D-sugar (denoted by the arrow). The linkage between the D and C units in Fondaparinux has this specific stereochemistry. There are, however, competing β- and α-glycosylation reactions.
The difficulties of the glycosylation reaction in the synthesis of Fondaparinux is well known. In 1991 Sanofi reported a preparation of a disaccharide intermediate in 51% yield having a 12/1 ratio of β/α stereochemistry at the anomeric position (Duchaussoy et al., Bioorg. & Med. Chem. Lett., 1(2), 99-102, 1991).
In another publication (Sinay et al, Carbohydrate Research, 132, C5-C9, 1984) yields on the order of 50% with coupling times on the order of 6- days are reported. U.S. Patent No. 4,818,816 {see e.g., column 31, lines 50-56) discloses a 50% yield for the β-glycosylation.
Alchemia’s U.S. Patent No. 7,541,445 is even less specific as to the details of the synthesis of this late-stage Fondaparinux synthetic intermediate. The ’445 Patent discloses several strategies for the assembly of the pentasaccharide (1+4, 3+2 or 2+3) using a 2-acylated D-sugar (specifically 2-allyloxycarbonyl) for the glycosylation coupling reactions. However, Alchemia’s strategy involves late-stage pentasaccharides that all incorporate a 2-benzylated D- sugar.
The transformation of acyl to benzyl is performed either under acidic or basic conditions. Furthermore, these transformations, using benzyl bromide or benzyl trichloroacetimidate, typically result in extensive decomposition and the procedure suffers from poor yields. Thus, such transformations (at a disaccharide, trisaccharide, and pentasaccharide level) are typically not acceptable for industrial scale production.
Examples of fully protected pentasaccharides are described in Duchaussoy et al, Bioorg. Med. Chem. Lett., 1 (2), 99-102, 1991; Petitou et al, Carbohydr. Res., 167, 67-75, 1987; Sinay et al, Carbohydr. Res., 132, C5-C9, 1984; Petitou et al., Carbohydr. Res., 1147, 221-236, 1986; Lei et al., Bioorg. Med. Chem., 6, 1337-1346, 1998; Ichikawa et al., Tet. Lett., 27(5), 611-614, 1986; Kovensky et al,Bioorg. Med. Chem., 1999, 7, 1567-1580, 1999.
These fully protected pentasaccharides may be converted to the O- and N-sulfated pentasaccharides using the four steps (described earlier) of: a) saponification with LiOHZH2CVNaOH, b) O-sulfation by an Et3N- SO3 complex; c) de-benzylation and azide reduction via H2/Pd hydrogenation; and d) N-sulfation with a pyridine-SO3 complex.
Even though many diverse analogs of the fully protected pentasaccharide have been prepared, none use any protective group at the 2-position of the D unit other than a benzyl group. Furthermore, none of the fully protected pentasaccharide analogs offer a practical, scaleable and economical method for re-introduction of the benzyl moiety at the 2-position of the D unit after removal of any participating group that promotes β-glycosylation.
Furthermore, the coupling of benzyl protected sugars proves to be a sluggish, low yielding and problematic process, typically resulting in substantial decomposition of the pentasaccharide (prepared over 50 synthetic steps), thus making it unsuitable for a large [kilogram] scale production process.
Ref. 1. Sinay et al, Carbohydr. Res., 132, C5-C9, 1984.
Ref. 2. Petitou et al., Carbohydr. Res., 147, 221-236. 1986
It has been a general strategy for carbohydrate chemists to use base-labile ester-protecting group at 2-position of the D unit to build an efficient and stereoselective β-glycosidic linkage. To construct the β-linkage carbohydrate chemists have previously acetate and benzoate ester groups, as described, for example, in the review by Poletti et al., Eur. J. Chem., 2999-3024, 2003.
The ester group at the 2-position of D needs to be differentiated from the acetate and benzoates at other positions in the pentasaccharide. These ester groups are hydrolyzed and sulfated later in the process and, unlike these ester groups, the 2-hydroxyl group of the D unit needs to remain as the hydroxyl group in the final product, Fondaparinux sodium.
Some of the current ester choices for the synthetic chemists in the field include methyl chloro acetyl and chloro methyl acetate [MCA or CMA] . The mild procedures for the selective removal of theses groups in the presence of acetates and benzoates makes them ideal candidates. However, MCA/CMA groups have been shown to produce unwanted and serious side products during the glycosylation and therefore have not been favored in the synthesis of Fondaparinux sodium and its analogs. For by-product formation observed in acetate derivatives see Seeberger et al., J. Org. Chem., 2004, 69, 4081-93.
Similar by-product formation is also observed using chloroacetate derivatives. See Orgueira et al., Eur. J. Chem., 9(1), 140-169, 2003.
The levulinyl group can be rapidly and almost quantitatively removed by treatment with hydrazine hydrate as the deprotection reagent as illustrated in the example below. Under the same reaction conditions primary and secondary acetate and benzoate esters are hardly affected by hydrazine hydrate. See, e.g., Seeberger et al, J. Org. Chem., 69, 4081-4093, 2004.
The syntheses of Fondaparinux sodium described herein takes advantage of the levulinyl group in efficient construction of the trisaccharide EDC with improved β- selectivity for the coupling under milder conditions and increased yields.
Substitution of the benzyl protecting group with a THP moiety provides its enhanced ability to be incorporated quantitatively in position-2 of the unit D of the pentasaccharide. Also, the THP group behaves in a similar manner to a benzyl ether in terms of function and stability. In the processes described herein, the THP group is incorporated at the 2-position of the D unit at this late stage of the synthesis (i.e., after the D and C units have been coupled through a 1,2-trans glycosidic (β-) linkage). The THP protective group typically does not promote an efficient β- glycosylation and therefore is preferably incorporated in the molecule after the construction of the β-linkage.
Fondaparinux and sodium salt thereof can be prepared from pure compound of Formula II by following the teachings from Bioorganic and Medicinal Chemistry Letters, 1(2), p. 95-98 (1991). A second aspect of the present invention provides a process for the preparation of 4-0- -D-glucopyranosyl-l,6-anhydro- -D-glucopyranose, represented by STR BELOW
……………………………..
SYNTHESIS
EP2464668A2 AND US8288515
The scheme below exemplifies some of the processes of the present invention disclosed herein.
The tetrahydropyranyl (THP) protective group and the benzyl ether protective group are suitable hydroxyl protective groups and can survive the last four synthetic steps (described above) in the synthesis of Fondaparinux sodium, even under harsh reaction conditions. Certain other protecting groups do not survive the last four synthetic steps in high yield.
Synthesis of Fondaparinux
Fondaparinux was prepared using the following procedure:

Synthetic Procedures
The following abbreviations are used herein: Ac is acetyl; ACN is acetonitrile; MS is molecular sieves; DMF is dimethyl formamide; PMB is p-methoxybenzyl; Bn is benzyl; DCM is dichloromethane; THF is tetrahydrofuran; TFA is trifluoro acetic acid; CSA is camphor sulfonic acid; TEA is triethylamine; MeOH is methanol; DMAP is dimethylaminopyridine; RT is room temperature; CAN is ceric ammonium nitrate; Ac2O is acetic anhydride; HBr is hydrogen bromide; TEMPO is tetramethylpiperidine-N-oxide; TBACl is tetrabutyl ammonium chloride; EtOAc is ethyl acetate; HOBT is hydroxybenzotriazole; DCC is dicyclohexylcarbodiimide; Lev is levunlinyl; TBDPS is tertiary-butyl diphenylsilyl; TCA is trichloroacetonitrile; O-TCA is O-trichloroacetimidate; Lev2O is levulinic anhydride; DIPEA is diisopropylethylamine; Bz is benzoyl; TBAF is tetrabutylammonium fluoride; DBU is diazabicycloundecane; BF3.Et2O is boron trifluoride etherate; TMSI is trimethylsilyl iodide; TBAI is tetrabutylammonium iodide; TES-Tf is triethylsilyl trifluoromethanesulfonate (triethylsilyl triflate); DHP is dihydropyran; PTS is p-toluenesulfonic acid.
Synthesis of Fondaparinux
Fondaparinux was prepared using the following procedure:
The ester moieties in EDCBA Pentamer were hydrolyzed with sodium and lithium hydroxide in the presence of hydrogen peroxide in dioxane mixing at room temperature for 16 hours to give the pentasaccharide intermediate API1. The five hydroxyl moieties in API1 were sulfated using a pyridine-sulfur trioxide complex in dimethylformamide, mixing at 60° C. for 2 hours and then purified using column chromatography (CG-161), to give the pentasulfated pentasaccharide API2. The intermediate API2 was then hydrogenated to reduce the three azides on sugars E, C and A to amines and the reductive deprotection of the five benzyl ethers to their corresponding hydroxyl groups to form the intermediate API3. This transformation occurs by reacting API2 with 10% palladium/carbon catalyst with hydrogen gas for 72 hours. The three amines on API3 were then sulfated using the pyridine-sulfur trioxide complex in sodium hydroxide and ammonium acetate, allowing the reaction to proceed for 12 hours. The acidic work-up procedure of the reaction removes the THP group to provide crude fondaparinux which is purified and is subsequently converted to its salt form. The crude mixture was purified using an ion-exchange chromatographic column (HiQ resin) followed by desalting using a size exclusion resin or gel filtration (Biorad Sephadex G25) to give the final API, fondaparinux sodium
Experimental Procedures Preparation of IntD1 Bromination of Glucose Pentaacetate
To a 500 ml flask was added 50 g of glucose pentaacetate (C6H22O11) and 80 ml of methylene chloride. The mixture was stirred at ice-water bath for 20 min HBr in HOAc (33%, 50 ml) was added to the reaction mixture. After stirring for 2.5 hr another 5 ml of HBr was added to the mixture. After another 30 min, the mixture was added 600 ml of methylene chloride. The organic mixture was washed with cold water (200 ml×2), Saturated NaHCO3(200 ml×2), water (200 ml) and brine (200 ml×2). The organic layer was dried over Na2SO4 and the mixture was evaporated at RT to give white solid as final product, bromide derivative, IntD1 (˜95% yield). C14H19BrO9, TLC Rf=0.49, SiO2, 40% ethyl acetate/60% hexanes; Exact Mass 410.02.
Preparation of IntD2 by Reductive Cyclization
To a stirring mixture of bromide IntD1 (105 g), tetrabutylammonium iodide (60 g, 162 mmol) and activated 3 Å molecular sieves in anhydrous acetonitrile (2 L), solid NaBH4 (30 g, 793 mmol) was added. The reaction was heated at 40° C. overnight. The mixture was then diluted with dichloromethane (2 L) and filtered through Celite®. After evaporation, the residue was dissolved in 500 ml ethyl acetate. The white solid (Bu4NI or Bu4NBr) was filtered. The ethyl acetate solution was evaporated and purified by chromatography on silica gel using ethyl acetate and hexane as eluent to give the acetal-triacetate IntD2 (˜60-70% yield). TLC Rf=0.36, SiO2 in 40% ethyl acetate/60% hexanes.
Preparation of IntD3 by De-Acetylation
To a 1000 ml flask was added triacetate IntD2 (55 g) and 500 ml of methanol. After stirring 30 min, the reagent NaOMe (2.7 g, 0.3 eq) was added and the reaction was stirred overnight. Additional NaOMe (0.9 g) was added to the reaction mixture and heated to 50° C. for 3 hr. The mixture was neutralized with Dowex 50Wx8 cation resin, filtered and evaporated. The residue was purified by silica gel column to give 24 g of trihydroxy-acetal IntD3. TLC Rf=0.36 in SiO2, 10% methanol/90% ethyl acetate.
Preparation of IntD4 by Benzylidene Formation
To a 1000 ml flask was added trihydroxy compound IntD3 (76 g) and benzaldehyde dimethyl acetate (73 g, 1.3 eq). The mixture was stirred for 10 min, after which D(+)-camphorsulfonic acid (8.5 g, CSA) was added. The mixture was heated at 50° C. for two hours. The reaction mixture was then transferred to separatory funnel containing ethyl acetate (1.8 L) and sodium bicarbonate solution (600 ml). After separation, the organic layer was washed with a second sodium bicarbonate solution (300 ml) and brine (800 ml). The two sodium carbonate solutions were combined and extracted with ethyl acetate (600 ml×2). The organic mixture was evaporated and purified by silica gel column to give the benzylidene product IntD4 (77 g, 71% yield). TLC Rf=0.47, SiO2 in 40% ethyl acetate/60% hexanes.
Preparation of IntD5 by Benzylation
To a 500 ml flask was added benzylidene acetal compound IntD4 (21 g,) in 70 ml THF. To another flask (1000 ml) was added NaH (2 eq). The solution of IntD4 was then transferred to the NaH solution at 0° C. The reaction mixture was stirred for 30 min, then benzyl bromide (16.1 ml, 1.9 eq) in 30 ml THF was added. After stirring for 30 min, DMF (90 ml) was added to the reaction mixture. Excess NaH was neutralized by careful addition of acetic acid (8 ml). The mixture was evaporated and purified by silica gel column to give the benzyl derivative IntD5. (23 g) TLC Rf=0.69, SiO2 in 40% ethyl acetate/60% hexanes.
Preparation of IntD6 by Deprotection of Benzylidene
To a 500 ml flask was added the benzylidene-acetal compound IntD5 (20 g) and 250 ml of dichloromethane, the reaction mixture was cooled to 0° C. using an ice-water-salt bath. Aqueous TFA (80%, 34 ml) was added to the mixture and stirred over night. The mixture was evaporated and purified by silica gel column to give the dihydroxy derivative IntD6. (8 g, 52%). TLC Rf=0.79, SiO2 in 10% methanol/90% ethyl acetate.
Preparation of IntD7 by Oxidation of 6-Hydroxyl
To a 5 L flask was added dihydroxy compound IntD6 (60 g), TEMPO (1.08 g), sodium bromide (4.2 g), tetrabutylammonium chloride (5.35 g), saturated NaHCO3 (794 ml) and EtOAc (1338 ml). The mixture was stirred over an ice-water bath for 30 min To another flask was added a solution of NaOCl (677 ml), saturated NaHCO3 (485 ml) and brine (794 ml). The second mixture was added slowly to the first mixture (over about two hrs). The resulting mixture was then stirred overnight. The mixture was separated, and the inorganic layer was extracted with EtOAc (800 ml×2). The combined organic layers were washed with brine (800 ml). Evaporation of the organic layer gave 64 g crude carboxylic acid product IntD7 which was used in the next step use without purification. TLC Rf=0.04, SiO2 in 10% methanol/90% ethyl acetate.
Preparation of Monomer D by Benzylation of the Carboxylic Acid
To a solution of carboxylic acid derivative IntD7 (64 g) in 600 ml of dichloromethane, was added benzyl alcohol (30 g) and N-hydroxybenzotriazole (80 g, HOBt). After stirring for 10 min triethylamine (60.2 g) was added slowly. After stirring another 10 min, dicyclohexylcarbodiimide, (60.8 g, DCC) was added slowly and the mixture was stirred overnight. The reaction mixture was filtered and the solvent was removed under reduced pressure followed by chromatography on silica gel to provide 60.8 g (75%, over two steps) of product, Monomer D. TLC Rf=0.51, SiO2 in 40% ethyl acetate/60% hexanes.
Synthesis of the BA Dimer
Step 1. Preparation of BMod1, Levulination of Monomer B1
A 100 L reactor was charged with 7.207 Kg of Monomer B1 (21.3 moles, 1 equiv), 20 L of dry tetrahydrofuran (THF) and agitated to dissolve. When clear, it was purged with nitrogen and 260 g of dimethylamino pyridine (DMAP, 2.13 moles, 0.1 equiv) and 11.05 L of diisopropylethylamine (DIPEA, 8.275 kg, 63.9 moles, 3 equiv) was charged into the reactor. The reactor was chilled to 10-15° C. and 13.7 kg levulinic anhydride (63.9 mol, 3 equiv) was transferred into the reactor. When the addition was complete, the reaction was warmed to ambient temperature and stirred overnight or 12-16 hours. Completeness of the reaction was monitored by TLC (40:60 ethyl acetate/hexane) and HPLC. When the reaction was complete, 20 L of 10% citric acid, 10 L of water and 25 L of ethyl acetate were transferred into the reactor. The mixture was stirred for 30 min and the layers were separated. The organic layer (EtOAc layer) was extracted with 20 L of water, 20 L 5% sodium bicarbonate and 20 L 25% brine solutions. The ethyl acetate solution was dried in 4-6 Kg of anhydrous sodium sulfate. The solution was evaporated to a syrup (bath temp. 40° C.) and dried overnight. The yield of the isolated syrup of BMod1 was 100%.
Synthesis of the BA Dimer
Step 2. Preparation of BMod2, TFA Hydrolysis of BMod1
A 100 L reactor was charged with 9296 Kg of 4-Lev Monomer B1 (BMod1) (21.3 mol, 1 equiv). The reactor chiller was turned to <5° C. and stirring was begun, after which 17.6 L of 90% TFA solution (TFA, 213 mole, 10 equiv) was transferred into the reactor. When the addition was complete, the reaction was monitored by TLC and HPLC. The reaction took approximately 2-3 hours to reach completion. When the reaction was complete, the reactor was chilled and 26.72 L of triethylamine (TEA, 19.4 Kg, 191.7 mole, 0.9 equiv) was transferred into the reactor. An additional 20 L of water and 20 L ethyl acetate were transferred into the reactor. This was stirred for 30 min and the layers were separated. The organic layer was extracted (EtOAc layer) with 20 L 5% sodium bicarbonate and 20 L 25% brine solutions. The ethyl acetate solution was dried in 4-6 Kg of anhydrous sodium sulfate. The solution was evaporated to a syrup (bath temp. 40° C.). The crude product was purified in a 200 L silica column using 140-200 L each of the following gradient profiles: 50:50, 80:20 (EtOAc/heptane), 100% EtOAc, 5:95, 10:90 (MeOH/EtOAc). The pure fractions were pooled and evaporated to a syrup. The yield of the isolated syrup, BMod2 was 90%.
Synthesis of the BA Dimer
Step 3. Preparation of BMod3, Silylation of BMod2
A 100 L reactor was charged with 6.755 Kg 4-Lev-1,2-DiOH Monomer B1 (BMod2) (17.04 mol, 1 equiv), 2328 g of imidazole (34.2 mol, 2 equiv) and 30 L of dichloromethane. The reactor was purged with nitrogen and chilled to −20° C., then 5.22 L tert-butyldiphenylchloro-silane (TBDPS-Cl, 5.607 Kg, 20.4 mol, 1.2 equiv) was transferred into the reactor. When addition was complete, the chiller was turned off and the reaction was allowed to warm to ambient temperature. The reaction was monitored by TLC (40% ethyl acetate/hexane) and HPLC. The reaction took approximately 3 hours to reach completion. When the reaction was complete, 20 L of water and 10 L of DCM were transferred into the reactor and stirred for 30 min, after which the layers were separated. The organic layer (DCM layer) was extracted with 20 L water and 20 L 25% brine solutions. Dichloromethane solution was dried in 4-6 Kg of anhydrous sodium sulfate. The solution was evaporated to a syrup (bath temp. 40° C.). The yield of BMod3 was about 80%.
Synthesis of the BA Dimer
Step 4. Preparation of BMod4, Benzoylation
A 100 L reactor was charged with 8.113 Kg of 4-Lev-1-Si-2-OH Monomer B1 (BMod3) (12.78 mol, 1 equiv), 9 L of pyridine and 30 L of dichloromethane. The reactor was purged with nitrogen and chilled to −20° C., after which 1.78 L of benzoyl chloride (2155 g, 15.34 mol, 1.2 equiv) was transferred into the reactor. When addition was complete, the reaction was allowed to warm to ambient temperature. The reaction was monitored by TLC (40% ethyl acetate/heptane) and HPLC. The reaction took approximately 3 hours to reach completion. When the reaction was complete, 20 L of water and 10 L of DCM were transferred into the reactor and stirred for 30 min, after which the layers were separated. The organic layer (DCM layer) was extracted with 20 L water and 20 L 25% brine solutions. The DCM solution was dried in 4-6 Kg of anhydrous sodium sulfate. The solution was evaporated to a syrup (bath temp. 40° C.). Isolated syrup BMod4 was obtained in 91% yield.
Synthesis of the BA Dimer
Step 5. Preparation of BMod5, Desilylation
A 100 L reactor was charged with 8.601 Kg of 4-Lev-1-Si-2-Bz Monomer B1 (BMod4) (11.64 mol, 1 equiv) in 30 L terahydrofuran. The reactor was purged with nitrogen and chilled to 0° C., after which 5.49 Kg of tetrabutylammonium fluoride (TBAF, 17.4 mol, 1.5 equiv) and 996 mL (1045 g, 17.4 mol, 1.5 equiv) of glacial acetic acid were transferred into the reactor. When the addition was complete, the reaction was stirred at ambient temperature. The reaction was monitored by TLC (40:60 ethyl acetate/hexane) and HPLC. The reaction took approximately 6 hours to reach completion. When the reaction was complete, 20 L of water and 10 L of DCM were transferred into the reactor and stirred for 30 min, after which the layers were separated. The organic layer (DCM layer) was extracted with 20 L water and 20 L 25% brine solutions. The dichloromethane solution was dried in 4-6 Kg of anhydrous sodium sulfate. The solution was evaporated to a syrup (bath temp. 40° C.). The crude product was purified in a 200 L silica column using 140-200 L each of the following gradient profiles: 10:90, 20:80, 30:70, 40:60, 50:50, 60:40, 70:30, 80:20 (EtOAc/heptane) and 200 L 100% EtOAc. Pure fractions were pooled and evaporated to a syrup. The intermediate BMod5 was isolated as a syrup in 91% yield.
Synthesis of the BA Dimer
Step 6: Preparation of BMod6, TCA Formation
A 100 L reactor was charged with 5.238 Kg of 4-Lev-1-OH-2-Bz Monomer B1 (BMod5) (10.44 mol, 1 equiv) in 30 L of DCM. The reactor was purged with nitrogen and chilled to 10-15° C., after which 780 mL of diazabicyclo undecene (DBU, 795 g, 5.22 mol, 0.5 equiv) and 10.47 L of trichloroacetonitrile (TCA, 15.08 Kg, 104.4 mol, 10 equiv) were transferred into the reactor. Stirring was continued and the reaction was kept under a nitrogen atmosphere. After reagent addition, the reaction was allowed to warm to ambient temperature. The reaction was monitored by HPLC and TLC (40:60 ethyl acetate/heptane). The reaction took approximately 2 hours to reach completion. When the reaction was complete, 20 L of water and 10 L of dichloromethane were transferred into the reactor. This was stirred for 30 min and the layers were separated. The organic layer (DCM layer) was separated with 20 L water and 20 L 25% brine solutions. The dichloromethane solution was dried in 4-6 Kg of anhydrous sodium sulfate. The solution was evaporated to a syrup (bath temp. 40° C.). The crude product was purified in a 200 L silica column using 140-200 L each of the following gradient profiles: 10:90, 20:80, 30:70, 40:60 and 50:50 (EtOAc/Heptane). Pure fractions were pooled and evaporated to a syrup. The isolated yield of BMod6 was 73%.
Synthesis of the BA Dimer
Step 7. Preparation of AMod1, Acetylation of Monomer A2
A 100 L reactor was charged with 6.772 Kg of Monomer A2 (17.04 mole, 1 eq.), 32.2 L (34.8 Kg, 340.8 moles, 20 eq.) of acetic anhydride and 32 L of dichloromethane. The reactor was purged with nitrogen and chilled to −20° C. When the temperature reached −20° C., 3.24 L (3.63 Kg, 25.68 mol, 1.5 equiv) of boron trifluoride etherate (BF3.Et2O) was transferred into the reactor. After complete addition of boron trifluoride etherate, the reaction was allowed to warm to room temperature. The completeness of the reaction was monitored by HPLC and TLC (30:70 ethyl acetate/heptane). The reaction took approximately 3-5 hours for completion. When the reaction was complete, extraction was performed with 3×15 L of 10% sodium bicarbonate and 20 L of water. The organic phase (DCM) was evaporated to a syrup (bath temp. 40° C.) and allowed to dry overnight. The syrup was purified in a 200 L silica column using 140 L each of the following gradient profiles: 5:95, 10:90, 20:80, 30:70, 40:60 and 50:50 (EtOAc/heptane). Pure fractions were pooled and evaporated to a syrup. The isolated yield of AMod1 was 83%.
Synthesis of the BA Dimer
Step 8. Preparation of AMod3,1-Methylation of AMod1
A 100 L reactor was charged with 5891 g of acetyl Monomer A2 (AMod1) (13.98 mole, 1 eq.) in 32 L of dichloromethane. The reactor was purged with nitrogen and was chilled to 0° C., after which 2598 mL of trimethylsilyl iodide (TMSI, 3636 g, 18 mol, 1.3 equiv) was transferred into the reactor. When addition was complete, the reaction was allowed to warm to room temperature. The completeness of the reaction was monitored by HPLC and TLC (30:70 ethyl acetate/heptane). The reaction took approximately 2-4 hours to reach completion. When the reaction was complete, the mixture was diluted with 20 L of toluene. The solution was evaporated to a syrup and was co-evaporated with 3×6 L of toluene. The reactor was charged with 36 L of dichloromethane (DCM), 3.2 Kg of dry 4 Å Molecular Sieves, 15505 g (42 mol, 3 equiv) of tetrabutyl ammonium iodide (TBAI) and 9 L of dry methanol. This was stirred until the TBAI was completely dissolved, after which 3630 mL of diisopropyl-ethylamine (DIPEA, 2712 g, 21 moles, 1.5 equiv) was transferred into the reactor in one portion. The completion of the reaction was monitored by HPLC and TLC (30:70 ethyl acetate/heptane). The reaction took approximately 16 hours for completion. When the reaction was complete, the molecular sieves were removed by filtration. Added were 20 L EtOAc and extracted with 4×20 L of 25% sodium thiosulfate and 20 L 10% NaCl solutions. The organic layer was separated and dried with 8-12 Kg of anhydrous sodium sulfate. The solution was evaporated to a syrup (bath temp. 40° C.). The crude product was purified in a 200 L silica column using 140-200 L each of the following gradient profiles: 5:95, 10:90, 20:80, 30:70 and 40:60 (EtOAc/heptane). The pure fractions were pooled and evaporated to give intermediate AMod3 as a syrup. The isolated yield was 75%.
Synthesis of the BA Dimer
Step 9. Preparation of AMod4, DeAcetylation of AMod3
A 100 L reactor was charged with 4128 g of 1-Methyl 4,6-Diacetyl Monomer A2 (AMod3) (10.5 mol, 1 equiv) and 18 L of dry methanol and dissolved, after which 113.4 g (2.1 mol, 0.2 equiv) of sodium methoxide was transferred into the reactor. The reaction was stirred at room temperature and monitored by TLC (40% ethyl acetate/hexane) and HPLC. The reaction took approximately 2-4 hours for completion. When the reaction was complete, Dowex 50Wx8 cation resin was added in small portions until the pH reached 6-8. The Dowex 50Wx8 resin was filtered and the solution was evaporated to a syrup (bath temp. 40° C.). The syrup was diluted with 10 L of ethyl acetate and extracted with 20 L brine and 20 L water. The ethyl acetate solution was dried in 4-6 Kg of anhydrous sodium sulfate. The solution was evaporated to a syrup (bath temp. 40° C.) and dried overnight at the same temperature. The isolated yield of the syrup AMod4 was about 88%.
Synthesis of the BA Dimer
Step 10. Preparation of AMod5,6-Benzoylation
A 100 L reactor was charged with 2858 g of Methyl 4,6-diOH Monomer A2 (AMod4) (9.24 mol, 1 equiv) and co-evaporated with 3×10 L of pyridine. When evaporation was complete, 15 L of dichloromethane, 6 L of pyridine were transferred into the reactor and dissolved. The reactor was purged with nitrogen and chilled to −40° C. The reactor was charged with 1044 mL (1299 g, 9.24 mol, 1 equiv) of benzoyl chloride. When the addition was complete, the reaction was allowed to warm to −10° C. over a period of 2 hours. The reaction was monitored by TLC (60% ethyl acetate/hexane). When the reaction was completed, the solution was evaporated to a syrup (bath temp. 40° C.). This was co-evaporated with 3×15 L of toluene. The syrup was diluted with 40 L ethyl acetate. Extraction was carried out with 20 L of water and 20 L of brine solution. The Ethyl acetate solution was dried in 4-6 Kg of anhydrous sodium sulfate. The solution was evaporated to a syrup (bath temp. 40° C.). The crude product was purified in a 200 L silica column using 140-200 L each of the following gradient profiles: 5:95, 10:90, 20:80, 25:70 and 30:60 (EtOAc/heptane). The pure fractions were pooled and evaporated to a syrup. The isolated yield of the intermediate AMod5 was 84%.
Synthesis of the BA Dimer
Step 11. Preparation of BA1, Coupling of Amod5 with BMod6
A 100 L reactor was charged with 3054 g of methyl 4-Hydroxy-Monomer A2 (AMod5) from Step 10 (7.38 mol, 1 equiv) and 4764 g of 4-Lev-1-TCA-Monomer B1 (BMod6) from Step 6 (7.38 mol, 1 equiv). The combined monomers were dissolved in 20 L of toluene and co-evaporated at 40° C. Co evaporation was repeated with an additional 2×20 L of toluene, after which 30 L of dichloromethane (DCM) was transferred into the reactor and dissolved. The reactor was purged with nitrogen and was chilled to below −20° C. When the temperature was between −20° C. and −40° C., 1572 g (1404 mL, 11.12 moles, 1.5 equiv) of boron trifluoride etherate (BF3.Et2O) were transferred into the reactor. After complete addition of boron trifluoride etherate, the reaction was allowed to warm to 0° C. and stirring was continued. The completeness of the reaction was monitored by HPLC and TLC (40:70 ethyl acetate/heptane). The reaction required 3-4 hours to reach completion. When the reaction was complete, 926 mL (672 g, 6.64 mol, 0.9 equiv) of triethylamine (TEA) was transferred into the mixture and stirred for an additional 30 minutes, after which 20 L of water and 10 L of dichloromethane were transferred into the reactor. The solution was stirred for 30 min and the layers were separated. The organic layer (DCM layer) was separated with 2×20 L water and 20 L 25% 4:1 sodium chloride/sodium bicarbonate solution. The dichloromethane solution was dried in 4-6 Kg of anhydrous sodium sulfate. The solution was evaporated to a syrup (bath temp. 40° C.) and used in the next step. The isolated yield of the disaccharide BA1 was about 72%.
Synthesis of the BA Dimer
Step 12, Removal of Levulinate Methyl [(methyl 2-O-benzoyl-3-O-benzyl-α-L-Idopyranosyluronate)-(1→4)-2-azido-6-O-benzoyl-3-O-benzyl]-2-deoxy-α-D-glucopyranoside
A 100 L reactor was charged with 4.104 Kg of 4-Lev BA Dimer (BA1) (4.56 mol, 1 equiv) in 20 L of THF. The reactor was purged with nitrogen and chilled to −20 to −25° C., after which 896 mL of hydrazine hydrate (923 g, 18.24 mol, 4 equiv) was transferred into the reactor. Stirring was continued and the reaction was monitored by TLC (40% ethyl acetate/heptane) and HPLC. The reaction took approximately 2-3 hour for the completion, after which 20 L of 10% citric acid, 10 L of water and 25 L of ethyl acetate were transferred into the reactor. This was stirred for 30 min and the layers were separated. The organic layer (ETOAc layer) was extracted with 20 L 25% brine solutions. The ethyl acetate solution was dried in 4-6 Kg of anhydrous sodium sulfate. The solution was evaporated to a syrup (bath temp. 40° C.). The crude product was purified in a 200 L silica column using 140-200 L each of the following gradient profiles: 10:90, 20:80, 30:70, 40:60 and 50:50 (EtOAc/heptane). The pure fractions were pooled and evaporated to dryness. The isolated yield of the BA Dimer was 82%. Formula: C42H43N3O13; Mol. Wt. 797.80.
Synthesis of the EDC Trimer
Step 1. Preparation of EMod1, Acetylation
A 100 L reactor was charged with 16533 g of Monomer E (45 mole, 1 eq.), 21.25 L acetic anhydride (225 mole, 5 eq.) and 60 L of dichloromethane. The reactor was purged with nitrogen and was chilled to −10° C. When the temperature was at −10° C., 1.14 L (1277 g) of boron trifluoride etherate (BF3.Et2O, 9.0 moles, 0.2 eq) were transferred into the reactor. After the complete addition of boron trifluoride etherate, the reaction was allowed to warm to room temperature. The completeness of the reaction was monitored by TLC (30:70 ethyl acetate/heptane) and HPLC. The reaction took approximately 3-6 hours to reach completion. When the reaction was completed, the mixture was extracted with 3×50 L of 10% sodium bicarbonate and SOL of water. The organic phase (DCM) was evaporated to a syrup (bath temp. 40° C.) and allowed to dry overnight. The isolated yield of EMod1 was 97%.
Synthesis of the EDC Trimer
Step 2. Preparation of EMod2, De-Acetylation of Azidoglucose
A 100 L reactor was charged with 21016 g of 1,6-Diacetyl Monomer E (EMod1) (45 mole, 1 eq.), 5434 g of hydrazine acetate (NH2NH2.HOAc, 24.75 mole, 0.55 eq.) and 50 L of DMF (dimethyl formamide). The solution was stirred at room temperature and the reaction was monitored by TLC (30% ethyl acetate/hexane) and HPLC. The reaction took approximately 2-4 hours for completion. When the reaction was completed, 50 L of dichloromethane and 40 L of water were transferred into the reactor. This was stirred for 30 minutes and the layers were separated. This was extracted with an additional 40 L of water and the organic phase was dried in 6-8 Kg of anhydrous sodium sulfate. The solution was evaporated to a syrup (bath temp. 40° C.) and dried overnight at the same temperature. The syrup was purified in a 200 L silica column using 140-200 L each of the following gradient profiles: 20:80, 30:70, 40:60 and 50:50 (EtOAc/heptane). Pure fractions were pooled and evaporated to a syrup. The isolated yield of intermediate EMod2 was 100%.
Synthesis of the EDC Trimer
Step 3. Preparation of EMod3, Formation of 1-TCA
A 100 L reactor was charged with 12752 g of 1-Hydroxy Monomer E (EMod2) (30 mole, 1 eq.) in 40 L of dichloromethane. The reactor was purged with nitrogen and stirring was started, after which 2.25 L of DBU (15 moles, 0.5 eq.) and 15.13 L of trichloroacetonitrile (150.9 moles, 5.03 eq) were transferred into the reactor. Stirring was continued and the reaction was kept under nitrogen. After the reagent addition, the reaction was allowed to warm to ambient temperature. The reaction was monitored by TLC (30:70 ethyl acetate/Heptane) and HPLC. The reaction took approximately 2-3 hours to reach completion. When the reaction was complete, 40 L of water and 20 L of DCM were charged into the reactor. This was stirred for 30 min and the layers were separated. The organic layer (DCM layer) was extracted with 40 L water and the DCM solution was dried in 6-8 Kg of anhydrous sodium sulfate. The solution was evaporated to a syrup (bath temp. 40° C.). The crude product was purified in a 200 L silica column using 140-200 L each of the following gradient profiles: 10:90 (DCM/EtOAc/heptane), 20:5:75 (DCM/EtOAc/heptane) and 20:10:70 DCM/EtOAc/heptane). Pure fractions were pooled and evaporated to give Intermediate EMod3 as a syrup. Isolated yield was 53%.
Synthesis of the EDC Trimer
Step 4. Preparation of ED Dimer, Coupling of E-TCA with Monomer D
A 100 L reactor was charged with 10471 g of 6-Acetyl-1-TCA Monomer E (EMod3) (18.3 mole, 1 eq., FW: 571.8) and 6594 g of Monomer D (16.47 mole, 0.9 eq, FW: 400.4). The combined monomers were dissolved in 20 L toluene and co-evaporated at 40° C. This was repeated with co-evaporation with an additional 2×20 L of toluene, after which 60 L of dichloromethane (DCM) were transferred into the reactor and dissolved. The reactor was purged with nitrogen and was chilled to −40° C. When the temperature was between −30° C. and −40° C., 2423 g (2071 mL, 9.17 moles, 0.5 eq) of TES Triflate were transferred into the reactor. After complete addition of TES Triflate the reaction was allowed to warm and stirring was continued. The completeness of the reaction was monitored by HPLC and TLC (35:65 ethyl acetate/Heptane). The reaction required 2-3 hours to reach completion. When the reaction was completed, 2040 mL of triethylamine (TEA, 1481 g, 0.8 eq.) were transferred into the reactor and stirred for an additional 30 minutes. The organic layer (DCM layer) was extracted with 2×20 L 25% 4:1 sodium chloride/sodium bicarbonate solution. The dichloromethane solution was dried in 6-8 Kg of anhydrous sodium sulfate. The syrup was purified in a 200 L silica column using 140-200 L each of the following gradient profiles: 15:85, 20:80, 25:75, 30:70 and 35:65 (EtOAc/heptane). Pure fractions were pooled and evaporated to a syrup. The ED Dimer was obtained in 81% isolated yield.
Synthesis of the EDC Trimer
Step 5. Preparation of ED1 TFA, Hydrolysis of ED Dimer
A 100 L reactor was charged with 7.5 Kg of ED Dimer (9.26 mol, 1 equiv). The reactor was chilled to <5° C. and 30.66 L of 90% TFA solution (TFA, 370.4 mol, 40 equiv) were transferred into the reactor. When the addition was completed the reaction was allowed to warm to room temperature. The reaction was monitored by TLC (40:60 ethyl acetate/hexanes) and HPLC. The reaction took approximately 3-4 hours to reach completion. When the reaction was completed, was chilled and 51.6 L of triethylamine (TEA, 37.5 Kg, 370.4 mole) were transferred into the reactor, after which 20 L of water & 20 L ethyl acetate were transferred into the reactor. This was stirred for 30 min and the layers were separated. The organic layer (EtOAc layer) was extracted with 20 L 5% sodium bicarbonate and 20 L 25% brine solutions. Ethyl acetate solution was dried in 4-6 Kg of anhydrous sodium sulfate. The solution was evaporated to a syrup (bath temp. 40° C.). The crude product was purified in a 200 L silica column using 140-200 L each of the following gradient profiles: 20:80, 30:70, 40:60, 50:50, 60:40 (EtOAc/heptane). The pure fractions were pooled and evaporated to a syrup. Isolated yield of ED1 was about 70%.
Synthesis of the EDC Trimer
Step 6. Preparation of ED2, Silylation of ED1
A 100 L reactor was charged with 11000 g of 1,2-diOH ED Dimer (ED1) (14.03 mol, 1 equiv), 1910.5 g of imidazole (28.06 mol, 2 equiv) and 30 L of dichloromethane. The reactor was purged with nitrogen and chilled to −20° C., after which 3.53 L butyldiphenylchloro-silane (TBDPS-Cl, 4.628 Kg, 16.835 mol, 1.2 equiv) was charged into the reactor. When the addition was complete, the chiller was turned off and the reaction was allowed to warm to ambient temperature. The reaction was monitored by TLC (50% ethyl acetate/hexane) and HPLC. The reaction required 4-6 hours to reach completion. When the reaction was completed, 20 L of water and 10 L of dichloromethane were transferred into the reactor and stirred for 30 min and the layers were separated. The organic layer (DCM layer) was extracted with 20 L water and 20 L 25% brine solutions. Dichloromethane solution was dried in 4-6 Kg of anhydrous sodium sulfate. The solution was evaporated to a syrup (bath temp. 40° C.). Intermediate ED2 was obtained in 75% isolated yield.
Synthesis of the EDC Trimer
Step 7. Preparation of ED3, D-Levulination
A 100 L reactor was charged with 19800 g of 1-Silyl ED Dimer (ED2) (19.37 moles, 1 equiv) and 40 L of dry tetrahydrofuran (THF) and agitated to dissolve. The reactor was purged with nitrogen and 237 g of dimethylaminopyridine (DMAP, 1.937 moles, 0.1 equiv) and 10.05 L of diisopropylethylamine (DIPEA, 63.9 moles, 3 equiv) were transferred into the reactor. The reactor was chilled to 10-15° C. and kept under a nitrogen atmosphere, after which 12.46 Kg of levulinic anhydride (58.11 moles, 3 eq) was charged into the reactor. When the addition was complete, the reaction was warmed to ambient temperature and stirred overnight or 12-16 hours. The completeness of the reaction was monitored by TLC (40:60 ethyl acetate/hexane) and by HPLC. 20 L of 10% citric acid, 10 L of water and 25 L of ethyl acetate were transferred into the reactor. This was stirred for 30 min and the layers were separated. The organic layer (EtOAc layer) was extracted with 20 L of water, 20 L 5% sodium bicarbonate and 20 L 25% brine solutions. The ethyl acetate solution was dried in 6-8 Kg of anhydrous sodium sulfate. The solution was evaporated to a syrup (bath temp. 40° C.). The ED3 yield was 95%.
Synthesis of the EDC Trimer
Step 8. Preparation of ED4, Desilylation of ED3
A 100 L reactor was charged with 19720 g of 1-Silyl-2-Lev ED Dimer (ED3) (17.6 mol, 1 equiv) in 40 L of THF. The reactor was chilled to 0° C., after which 6903 g of tetrabutylammonium fluoride trihydrate (TBAF, 26.4 mol, 1.5 equiv) and 1511 mL (26.4 mol, 1.5 equiv) of glacial acetic acid were transferred into the reactor. When the addition was complete, the reaction was stirred and allowed to warm to ambient temperature. The reaction was monitored by TLC (40:60 ethyl acetate/hexane) and HPLC. The reaction required 3 hours to reach completion. When the reaction was completed, 20 L of water and 10 L of dichloromethane were transferred into the reactor and stirred for 30 min and the layers were separated. The organic layer (DCM layer) was extracted with 20 L water and 20 L 25% brine solutions. The dichloromethane solution was dried in 6-8 Kg of anhydrous sodium sulfate. The solution was evaporated to a syrup (bath temp. 40° C.). The crude product was purified using a 200 L silica column using 140-200 L each of the following gradient profiles: 10:90, 20:80, 30:70, 40:60, 50:50, 60:40, 70:30, 80:20 (EtOAc/heptane) and 200 L 100% EtOAc. The pure fractions were pooled and evaporated to a syrup and used in the next step. The isolated yield of ED4 was about 92%.
Synthesis of the EDC Trimer
Step 9. Preparation of ED5, TCA Formation
A 100 L reactor was charged with 14420 g of 1-OH-2-Lev ED Dimer (ED4) (16.35 mol, 1 equiv) in 30 L of dichloromethane. The reactor was purged with nitrogen and stirring was begun, after which 1222 mL of diazabicycloundecene (DBU, 8.175 mol, 0.5 equiv) and 23.61 Kg of trichloroacetonitrile (TCA, 163.5 mol, 10 equiv) were transferred into the reactor. Stirring was continued and the reaction was kept under nitrogen. After reagent addition, the reaction was allowed to warm to ambient temperature. The reaction was monitored by HPLC and TLC (40:60 ethyl acetate/heptane). The reaction took approximately 2 hours for reaction completion. When the reaction was completed, 20 L of water and 10 L of DCM were transferred into the reactor and stirred for 30 min and the layers were separated. The organic layer (DCM layer) was extracted with 20 L water and 20 L 25% brine solutions. The dichloromethane solution was dried in 6-8 Kg of anhydrous sodium sulfate. The solution was evaporated to a syrup (bath temp. 40° C.). The crude product was purified using a 200 L silica column using 140-200 L each of the following gradient profiles: 10:90, 20:80, 30:70, 40:60 and 50:50 (EtOAc/heptane). The pure fractions were pooled and evaporated to a syrup and used in the next step. The isolated yield of intermediate ED5 was about 70%.
Synthesis of the EDC Trimer
Step 10.
Preparation of EDC Trimer, Coupling of ED5 with Monomer C 6-O-acetyl-2-azido-3,4-di-O-benzyl-2-deoxy-α-D-glucopyranosyl-(1→4)-benzyl (3-O-benzyl-2-O-levulinoyl)-β-D-glucopyranosyluronate-(1→4)-(3-O-acetyl-1,6-anhydro-2-azido)-2-deoxy-β-D-glucopyranose
A 100 L reactor was charged with 12780 g of 2-Lev 1-TCA ED Dimer (ED5) (7.38 mole, 1 eq., FW) and 4764 g of Monomer C (7.38 mole, 1 eq). The combined monomers were dissolved in 20 L toluene and co-evaporated at 40° C. Repeated was co-evaporation with an additional 2×20 L of toluene, after which 60 L of dichloromethane (DCM) was transferred into the reactor and dissolved. The reactor was purged with nitrogen and chilled to −20° C. When the temperature was between −20 and −10° C., 2962 g (11.2 moles, 0.9 eq) of TES Triflate were transferred into the reactor. After complete addition of TES Triflate the reaction was allowed to warm to 5° C. and stirring was continued. Completeness of the reaction was monitored by HPLC and TLC (35:65 ethyl acetate/Heptane). The reaction required 2-3 hours to reach completion. When the reaction was completed, 1133 g of triethylamine (TEA, 0.9 eq.) were transferred into the reactor and stirred for an additional 30 minutes. The organic layer (DCM layer) was extracted with 2×20 L 25% 4:1 sodium chloride/sodium bicarbonate solution. Dichloromethane solution was dried in 6-8 Kg of anhydrous sodium sulfate. The syrup was purified in a 200 L silica column using 140-200 L each of the following gradient profiles: 15:85, 20:80, 25:75, 30:70 and 35:65 (EtOAc/heptane). Pure fractions were pooled and evaporated to a syrup. The isolated yield of EDC Trimer was 48%. Formula: C55H60N6O18; Mol. Wt. 1093.09. The 1H NMR spectrum (d6-acetone) of the EDC trimer is shown in FIG. 3.

Preparation of EDC1
Step 1:
Anhydro Ring Opening & Acetylation 6-O-acetyl-2-azido-2-deoxy-3,4-di-O-benzyl-α-D-glucopyranosyl-(1→4)-O-[benzyl 3-O-benzyl-2-O-levulinoyl-β-D-glucopyranosyluronate]-(1→4)-O-2-azido-2-deoxy-1,3,6-tri-O-acetyl-β-D-glucopyranose
7.0 Kg (6.44 mol) of EDC Trimer was dissolved in 18 L anhydrous Dichloromethane. 6.57 Kg (64.4 mol, 10 eq) of Acetic anhydride was added. The solution was cooled to −45 to −35° C. and 1.82 Kg (12.9 mol, 2 eq) of Boron Trifluoride etherate was added slowly. Upon completion of addition, the mixture was warmed to 0-10° C. and kept at this temperature for 3 hours until reaction was complete by TLC and HPLC. The reaction was cooled to −20° C. and cautiously quenched and extracted with saturated solution of sodium bicarbonate (3×20 L) while maintaining the mixture temperature below 5° C. The organic layer was extracted with brine (1×20 L), dried over anhydrous sodium sulfate, and concentrated under vacuum to a syrup. The resulting syrup of EDC1 (6.74 Kg) was used for step 2 without further purification. The 1H NMR spectrum (d6-acetone) of the EDC-1 trimer is shown in FIG. 4.

Preparation of EDC2
Step 2:
Deacetylation 6-O-acetyl-2-azido-2-deoxy-3,4-di-O-benzyl-α-D-glucopyranosyl-(1→4)-O-[benzyl 3-O-benzyl-2-O-levulinoyl-β-D-glucopyranosyluronate]-(1→4)-O-2-azido-2-deoxy-3,6-di-O-acetyl-β-D-glucopyranose
The crude EDC1 product obtained from step 1 was dissolved in 27 L of Tetrahydrofuran and chilled to 15-20° C., after which 6 Kg (55.8 mol) of benzylamine was added slowly while maintaining the reaction temperature below 25° C. The reaction mixture was stirred for 5-6 hours at 10-15° C. Upon completion, the mixture was diluted with ethyl acetate and extracted and quenched with 10% citric acid solution (2×20 L) while maintaining the temperature below 25° C. The organic layer was extracted with 10% NaCl/1% sodium bicarbonate (1×20 L). The extraction was repeated with water (1×10 L), dried over anhydrous sodium sulfate and evaporated under vacuum to a syrup. Column chromatographic separation using silica gel yielded 4.21 Kg (57% yield over 2 steps) of EDC2[ also referred to as 1-Hydroxy-6-Acetyl EDC Trimer]. The 1H NMR spectrum (d6-acetone) of the EDC-2 trimer is shown in FIG. 5.

Preparation of EDC3
Step 3:
Formation of TCA Derivative 6-O-acetyl-2-azido-2-deoxy-3,4-di-O-benzyl-α-D-glucopyranosyl-(1→4)-O-[benzyl 3-O-benzyl-2-O-levulinoyl-β-D-glucopyranosyluronate]-(1→4)-O-2-azido-2-deoxy-3,6-di-O-acetyl-1-O-trichloroacetimidoyl-β-D-glucopyranose
4.54 Kg (3.94 mol) of EDC2 was dissolved in 20 L of Dichloromethane. 11.4 Kg (78.8 mol, 20 eq) of Trichloroacetonitrile was added. The solution was cooled to −15 to −20° C. and 300 g (1.97 mol, 0.5 eq) of Diazabicycloundecene was added. The reaction was allowed to warm to 0-10° C. and stirred for 2 hours or until reaction was complete. Upon completion, water (20 L) was added and the reaction was extracted with an additional 10 L of DCM. The organic layer was extracted with brine (1×20 L), dried over anhydrous sodium sulfate, and concentrated under vacuum to a syrup. Column chromatographic separation using silica gel and 20-60% ethyl acetate/heptane gradient yielded 3.67 Kg (72% yield) of 1-TCA derivative, EDC3. The 1H NMR spectrum (d6-acetone) of the EDC-3 trimer is shown in FIG. 6.

Preparation of EDCBA1
Step 4:
Coupling of EDC3 with BA Dimer Methyl O-6-O-acetyl-2-azido-2-deoxy-3,4-di-O-benzyl-α-D-glucopyranosyl)-(1→4)-O-[benzyl 3-O-benzyl-2-O-levulinoyl-β-D-glucopyranosyluronate]-(1→4)-O-2-azido-2-deoxy-3,6-di-O-acetyl-α-D-glucopyranosyl-(1→4)-O-[methyl 2-O-benzoyl-3-O-benzyl-α-L-Idopyranosyluronate]-(1→4)-2-azido-6-O-benzoyl-3-O-benzyl-2-deoxy-α-D-glucopyranoside
3.67 Kg (2.83 mol) of EDC3 and 3.16 Kg (3.96 mol, 1.4 eq) of BA Dimer was dissolved in 7-10 L of Toluene and evaporated to dryness. The resulting syrup was coevaporated with Toluene (2×15 L) to remove water. The dried syrup was dissolved in 20 L of anhydrous Dichloromethane, transferred to the reaction flask, and cooled to −15 to −20° C. 898 g (3.4 mol, 1.2 eq) of triethylsilyl triflate was added while maintaining the temperature below −5° C. When the addition was complete, the reaction was immediately warmed to −5 to 0° C. and stirred for 3 hours. The reaction was quenched by slowly adding 344 g (3.4 mol, 1.2 eq) of Triethylamine. Water (15 L) was added and the reaction was extracted with an additional 10 L of DCM. The organic layer was extracted with a 25% 4:1 Sodium Chloride/Sodium Bicarbonate solution (2×20 L), dried over anhydrous sodium sulfate, and evaporated under vacuum to a syrup. The resulting syrup of the pentasaccharide, EDCBA1 was used for step 5 without further purification. The 1H NMR spectrum (d6-acetone) of the EDCBA-1 pentamer is shown in FIG. 7.

Preparation of EDCBA2
Step 5:
Hydrolysis of Levulinyl moiety Methyl O-6-O-acetyl-2-azido-2-deoxy-3,4-di-O-benzyl-α-D-glucopyranosyl)-(1→4)—O-[benzyl 3-O-benzyl-β-D-glucopyranosyluronate]-(1→4)-O-2-azido-2-deoxy-3,6-di-O-acetyl-α-D-glucopyranosyl)-(1→4)-O-[methyl 2-O-benzoyl-3-O-benzyl-α-L-Idopyranosyluronate]-(1→4)-2-azido-6-O-benzoyl-3-O-benzyl-2-deoxy-α-D-glucopyranoside
The crude EDCBA1 from step 4 was dissolved in 15 L of Tetrahydrofuran and chilled to −20 to −25° C. A solution containing 679 g (13.6 mol) of Hydrazine monohydrate and 171 g (1.94 mol) of Hydrazine Acetate in 7 L of Methanol was added slowly while maintaining the temperature below −20° C. When the addition was complete, the reaction mixture was allowed to warm to 0-10° C. and stirred for several hours until the reaction is complete, after which 20 L of Ethyl acetate was added and the reaction was extracted with 10% citric acid (2×12 L). The organic layer was washed with water (1×12 L), dried over anhydrous sodium sulfate, and evaporated under vacuum to a syrup. Column chromatographic separation using silica gel and 10-45% ethyl acetate/heptane gradient yielded 2.47 Kg (47.5% yield over 2 steps) of EDCBA2. The 1H NMR spectrum (d6-acetone) of the EDCBA-2 pentamer is shown in FIG. 8.

Preparation of EDCBA Pentamer
Step 6:
THP Formation Methyl O-6-O-acetyl-2-azido-2-deoxy-3,4-di-O-benzyl-α-D-glucopyranosyl-(1→4)-O-[benzyl 3-O-benzyl-2-O-tetrahydropyranyl-β-D-glucopyranosyluronate]-(1→4)-O-2-azido-2-deoxy-3,6-di-O-acetyl-α-D-glucopyranosyl-(1→4)-O-[methyl 2-O-benzoyl-3-O-benzyl-α-L-Idopyranosyluronate]-(1→4)-2-azido-6-O-benzoyl-3-O-benzyl-2-deoxy-α-D-glucopyranoside
2.47 Kg (1.35 mol) of EDCBA2 was dissolved in 23 L Dichloroethane and chilled to 10-15° C., after which 1.13 Kg (13.5 mol, 10 eq) of Dihydropyran and 31.3 g (0.135 mol, 0.1 eq) of Camphorsulfonic acid were added. The reaction was allowed warm to 20-25° C. and stirred for 4-6 hours until reaction was complete. Water (15 L) was added and the reaction was extracted with an additional 10 L of DCE. The organic layer was extracted with a 25% 4:1 Sodium Chloride/Sodium Bicarbonate solution (2×20 L), dried over anhydrous sodium sulfate, and evaporated under vacuum to a syrup. Column chromatographic separation using silica gel and 10-35% ethyl acetate/heptane gradient yielded 2.28 Kg (88.5% yield) of fully protected EDCBA Pentamer. The 1H NMR spectrum (d6-acetone) of the EDCBA pentamer is shown in FIG. 9.

Preparation of API1
Step 1:
Saponification Methyl O-2-azido-2-deoxy-3,4-di-O-benzyl-α-D-glucopyranosyl-(1→4)-O-3-O-benzyl-2-O-tetrahydropyranyl-β-D-glucopyranosyluronosyl-(1→4)-O-2-azido-2-deoxy-α-D-glucopyranosyl-(1→4)-O-3-O-benzyl-α-L-Idopyranosyluronosyl-(1→4)-2-azido-3-O-benzyl-2-deoxy-α-D-glucopyranoside disodium salt
To a solution of 2.28 Kg (1.19 mol) of EDCBA Pentamer in 27 L of Dioxane and 41 L of Tetrahydrofuran was added 45.5 L of 0.7 M (31.88 mol, 27 eq) Lithium hydroxide solution followed by 5.33 L of 30% Hydrogen peroxide. The reaction mixture was stirred for 10-20 hours to remove the acetyl groups. Then, 10 L of 4 N (40 mol, 34 eq) sodium hydroxide solution was added. The reaction was allowed to stir for an additional 24-48 hours to hydrolyze the benzyl and methyl esters completely. The reaction was then extracted with ethyl acetate. The organic layer was extracted with brine solution and dried with anhydrous sodium sulfate. Evaporation of the solvent under vacuum gave a syrup of API1 [also referred to as EDCBA(OH)5] which was used for the next step without further purification.
Preparation of API2
Step 2:
O-Sulfonation Methyl O-2-azido-2-deoxy-3,4-di-O-benzyl-6-O-sulfo-α-D-glucopyranosyl-(1→4)-O-3-O-benzyl-2-O-tetrahydropyranyl-β-D-glucopyranosyluronosyl-(1→4)-O-2-azido-2-deoxy-3,6-di-O-sulfo-α-D-glucopyranosyl-(1→4)-O-3-O-benzyl-2-O-sulfo-α-L-idopyranuronosyl-(1→4)-2-azido-2-deoxy-6-O-sulfo-α-D-glucopyranoside, heptasodium salt
The crude product of API1 [aka EDCBA(OH)5] obtained in step 1 was dissolved in 10 L Dimethylformamide. To this was added a previously prepared solution containing 10.5 Kg (66 moles) of sulfur trioxide-pyridine complex in 10 L of Pyridine and 25 L of Dimethylformamide. The reaction mixture was heated to 50° C. over a period of 45 min. After stiffing at 1.5 hours at 50° C., the reaction was cooled to 20° C. and was quenched into 60 L of 8% sodium bicarbonate solution that was kept at 10° C. The pH of the quench mixture was maintained at pH 7-9 by addition of sodium bicarbonate solution. When all the reaction mixture has been transferred, the quench mixture was stirred for an additional 2 hours and pH was maintained at pH 7 or greater. When the pH of quench has stabilized, it was diluted with water and the resulting mixture was purified using a preparative HPLC column packed with Amberchrom CG161-M and eluted with 90%-10% Sodium Bicarbonate (5%) solution/Methanol over 180 min. The pure fractions were concentrated under vacuum and was then desalted using a size exclusion resin or gel filtration (Biorad) G25 to give 1581 g (65.5% yield over 2 steps) of API2 [also referred to as EDCBA(OSO3)5]. The 1H NMR spectrum (d6-acetone) of API-2 pentamer is shown in FIG. 10.

Preparation of API3
Step 3:
Hydrogenation Methyl O-2-amino-2-deoxy-6-O-sulfo-α-D-glucopyranosyl-(1→4)-O-2-O-tetrahydropyranyl-β-D-glucopyranosyluronosyl-(1→4)-O-2-amino-2-deoxy-3,6-di-O-sulfo-α-D-glucopyranosyl-(1→4)-O-2-O-sulfo-α-L-idopyranuronosyl-(1→4)-2-amino-2-deoxy-6-O-sulfo-α-D-glucopyranoside, heptasodium salt
A solution of 1581 g (0.78 mol) of O-Sulfated pentasaccharide API2 in 38 L of Methanol and 32 L of water was treated with 30 wt % of Palladium in Activated carbon under 100 psi of Hydrogen pressure at 60-65° C. for 60 hours or until completion of reaction. The mixture was then filtered through 1.0μ and 0.2μ filter cartridges and the solvent evaporated under vacuum to give 942 g (80% yield) of API3 [also referred to as EDCBA(OSO3)5(NH2)3]. The 1H NMR spectrum (d6-acetone) of API-3 pentamer is shown in FIG. 11.

Preparation of Fondaparinux Sodium
Step 4:
N-Sulfation & Removal of THP Methyl O-2-deoxy-6-O-sulfo-2-(sulfoamino)-α-D-glucopyranosyl-(1→4)—O-β-D-glucopyranuronosyl-(1→4)-O-2-deoxy-3,6-di-O-sulfo-2-(sulfoamino)-α-D-glucopyranosyl-(1→4)-O-2-O-sulfo-α-L-idopyranuronosyl-(1→4)-2-deoxy-6-O-sulfo-2-(sulfoamino)-α-D-glucopyranoside, decasodium salt
To a solution of 942 g (0.63 mol) of API3 in 46 L of water was slowly added 3.25 Kg (20.4 mol, 32 eq) of Sulfur trioxide-pyridine complex, maintaining the pH of the reaction mixture at pH 9-9.5 during the addition using 2 N sodium hydroxide solution. The reaction was allowed to stir for 4-6 hours at pH 9.0-9.5. When reaction was complete, the pH was adjusted to pH 7.0 using 50 mM solution of Ammonium acetate at pH 3.5. The resulting N-sulfated EDCBA(OSO3)5(NHSO3)3 mixture was purified using Ion-Exchange Chromatographic Column (Varian Preparative 15 cm HiQ Column) followed by desalting using a size exclusion resin or gel filtration (Biorad G25). The resulting mixture was then treated with activated charcoal and the purification by ion-exchange and desalting were repeated to give 516 g (47.6% yield) of the purified Fondaparinux Sodium form.
Analysis of the Fondaparinux sodium identified the presence of P1, P2, P3, and P4 in the fondaparinux. P1, P2, P3, and P4 were identified by standard analytical methods.
INTERMEDIATES
The monomers used in the processes described herein may be prepared as described in the art, or can be prepared using the methods described herein.
The synthesis of Monomer A-2 (CAS Registry Number 134221-42-4) has been described in the following references: Arndt et al., Organic Letters, 5(22), 4179-4182, 2003; Sakairi et al., Bulletin of the Chemical Society of Japan, 67(6), 1756-8, 1994; and Sakairi et al., Journal of the Chemical Society, Chemical Communications, (5), 289-90, 1991, and the references cited therein, which are hereby incorporated by reference in their entireties.
Monomer C(CAS Registry Number 87326-68-9) can be synthesized using the methods described in the following references: Ganguli et al., Tetrahedron: Asymmetry, 16(2), 411-424, 2005; Izumi et al., Journal of Organic Chemistry, 62(4), 992-998, 1997; Van Boeckel et al., Recueil: Journal of the Royal Netherlands Chemical Society, 102(9), 415-16, 1983; Wessel et al.,Helvetica Chimica Acta, 72(6), 1268-77, 1989; Petitou et al., U.S. Pat. No. 4,818,816 and references cited therein, which are hereby incorporated by reference in their entireties.
Monomer E (CAS Registry Number 55682-48-9) can be synthesized using the methods described in the following literature references: Hawley et al., European Journal of Organic Chemistry, (12), 1925-1936, 2002; Dondoni et al., Journal of Organic Chemistry, 67(13), 4475-4486, 2002; Van der Klein et al., Tetrahedron, 48(22), 4649-58, 1992; Hori et al., Journal of Organic Chemistry, 54(6), 1346-53, 1989; Sakairi et al., Bulletin of the Chemical Society of Japan, 67(6), 1756-8, 1994; Tailler et al.,Journal of the Chemical Society, Perkin Transactions 1: Organic and Bio-Organic Chemistry, (23), 3163-4, (1972-1999) (1992); Paulsen et al., Chemische Berichte, 111(6), 2334-47, 1978; Dasgupta et al., Synthesis, (8), 626-8, 1988; Paulsen et al., Angewandte Chemie, 87(15), 547-8, 1975; and references cited therein, which are hereby incorporated by reference in their entireties.
Monomer B-1 (CAS Registry Number 444118-44-9) can be synthesized using the methods described in the following literature references: Lohman et al., Journal of Organic Chemistry, 68(19), 7559-7561, 2003; Orgueira et al., Chemistry—A European Journal, 9(1), 140-169, 2003; Manabe et al., Journal of the American Chemical Society, 128(33), 10666-10667, 2006; Orgueira et al., Angewandte Chemie, International Edition, 41(12), 2128-2131, 2002; and references cited therein, which are hereby incorporated by reference in their entireties.
Synthesis of Monomer D
Monomer D was prepared in 8 synthetic steps from glucose pentaacetate using the following procedure:
Synthesis of Monomer D
Monomer D was prepared in 8 synthetic steps from glucose pentaacetate using the following procedure:
Pentaacetate SM-B was brominated at the anomeric carbon using HBr in acetic acid to give bromide derivative IntD1. This step was carried out using the reactants SM-B, 33% hydrogen bromide, acetic acid and dichloromethane, stirring in an ice water bath for about 3 hours and evaporating at room temperature. IntD1 was reductively cyclized with sodium borohydride and tetrabutylammonium iodide in acetonitrile using 3 Å molecular sieves as dehydrating agent and stirring at 40° C. for 16 hours to give the acetal derivative, IntD2. The three acetyl groups in IntD2 were hydrolyzed by heating with sodium methoxide in methanol at 50° C. for 3 hours and the reaction mixture was neutralized using Dowex 50WX8-100 resin (Aldrich) in the acid form to give the trihydroxy acetal derivative IntD3.
The C4 and C6 hydroxyls of IntD3 were protected by mixing with benzaldehyde dimethyl acetate and camphor sulphonic acid at 50° C. for 2 hours to give the benzylidene-acetal derivative IntD4. The free hydroxyl at the C3 position of IntD4 was deprotonated with sodium hydride in THF as solvent at 0° C. and alkylated with benzyl bromide in THF, and allowing the reaction mixture to warm to room temperature with stirring to give the benzyl ether IntD5. The benzylidene moiety of IntD5 was deprotected by adding trifluoroacetic acid in dichloromethane at 0° C. and allowing it to warm to room temperature for 16 hours to give IntD6 with a primary hydroxyl group. IntD6 was then oxidized with TEMPO (2,2,6,6-tetramethyl-1-piperidine-N-oxide) in the presence of tetrabutylammonium chloride, sodium bromide, ethyl acetate, sodium chlorate and sodium bicarbonate, with stirring at room temperature for 16 hours to form the carboxylic acid derivative IntD7. The acid IntD7 was esterified with benzyl alcohol and dicyclohexylcarbodiimide (other reactants being hydroxybenzotriazole and triethylamine) with stirring at room temperature for 16 hours to give Monomer D.
Synthesis of the BA Dimer
The BA Dimer was prepared in 12 synthetic steps from Monomer B1 and Monomer A2 using the following procedure:
The C4-hydroxyl of Monomer B-1 was levulinated using levulinic anhydride and diisopropylethylamine (DIPEA) with mixing at room temperature for 16 hours to give the levulinate ester BMod1, which was followed by hydrolysis of the acetonide with 90% trifluoroacetic acid and mixing at room temperature for 4 hours to give the diol BMod2. The C1 hydroxyl of the diol BMod2 was silylated with tert-butyldiphenylsilylchloride by mixing at room temperature for 3 hours to give silyl derivative BMod3. The C2-hydroxyl was then benzoylated with benzoyl chloride in pyridine, and mixed at room temperature for 3 hours to give compound BMod4. The silyl group on BMod4 was then deprotected with tert-butyl ammonium fluoride and mixing at room temperature for 3 hours to give the C1-hydroyl BMod5. The C1-hydroxyl is then allowed to react with trichloroacetonitrile in the presence of diazobicycloundecane (DBU) and mixing at room temperature for 2 hours to give the trichloroacetamidate (TCA) derivative BMod6, which suitable for coupling, for example with Monomer A-2.
Monomer A-2 was prepared for coupling by opening the anhydro moiety with BF3.Et2O followed by acetylation of the resulting hydroxyl groups to give the triacetate derivative AMod1.
Monomer A2 was prepared for the coupling reaction by opening the anhydro moiety and acetylation of the resulting hydroxyl groups to give the triacetate derivative AMod1. This transformation occurs using boron trifluoride etherate, acetic anhydride and dichloromethane, between −20° C. and room temperature for 3 hours. The C1-Acetate of AMod1 was then hydrolyzed and methylated in two steps to give the diacetate AMod3. That is, first AMod1 was reacted with trimethylsilyl iodide and mixed at room temperature for 2 hours, then reacted with and tetrabutyl ammonium iodide. This mixture was reacted with diisoproylethylamine and methanol and stirred for 16 hours at room temperature, thus forming AMod3. The C4 and C6 acetates of AMod3 are hydrolyzed with sodium methoxide to give the diol Amod4. The AMod3 mixture was also subjected to mixing at room temperature for 3 hours with Dowex 50 Wx4x8-100 resin in the acid form for neutralization. This formed Amod4. The C6-hydroxyl of AMod4 is then benzoylated by treating with benzoyl chloride in pyridine at −40° C. and then allowing it to warm up to −10° C. over 2 hours to give AMod5.
Coupling of monomer AMod5 with the free C4-hydroxyl group of BMod6 was performed in the presence of BF3.Et2O and dichloromethane with mixing between −20° C. and room temperature for 3 hours to provide disaccharide BA1. The C4-levulinyl moiety of the disaccharide was then hydrolyzed with hydrazine to give the BA Dimer, which is suitable for subsequent coupling reactions.
Synthesis of EDC Trimer
The EDC Trimer was prepared in 10 synthetic steps from Monomer E, Monomer D and Monomer C using the following procedure:
Monomer E was prepared for coupling by opening the anhydro moiety with BF3.Et2O followed by acetylation of the resulting hydroxyl groups to give diacetate EMod1. This occurs by the addition of Monomer E with boron trifluoride etherate, acetic anhydride and dichloromethane at −10° C., and allowing the reaction to warm to room temperature with stirring for 3 hours. The C1-Acetate of EMod1 is then hydrolyzed to give the alcohol, EMod2. This occurs by reacting Emod1 with hydrazine acetate and dimethylformamide and mixing at room temperature for 3 hours. The C1-hydroxyl of Emod2 is then reacted with trichloroacetonitrile to give the trichloro acetamidate (TCA) derivative EMod3 suitable for coupling, which reaction also employs diazabicycloundecane and dichloromethane and mixing at room temperature for 2 hours.
Monomer D, having a free C4-hydroxyl group, was coupled with monomer EMod3 in the presence of triethylsilyl triflate with mixing at −40° C. for 2 hours to give the disaccharide ED Dimer. The acetal on ring sugar D of the ED Dimer is hydrolyzed to give the C1,C2-diol ED1. This occurs by reacting the ED Dimer with 90% trifluoro acetic acid and mixing at room temperature for 4 hours. The C1-hydroxyl moiety of ED1 was then silylated with tert-butyldiphenylsilyl chloride to give the silyl derivative ED2. The C2-hydroxyl of ED2 was then allowed to react with levulinic anhydride in the presence of dimethylaminopyridine (DMAP) and diethylisopropylamine for approximately 16 hours to give the levulinate ester ED3. The TBDPS moiety is then deprotected by removal with tert-butylammonium fluoride in acetic acid with mixing at room temperature for 3 hours to give ED4 having a C1-hydroxyl. The C1-hydroxyl moiety of ED4 was then allowed to react with trichloroacetonitrile to give the TCA derivative ED5, which is suitable for coupling.
The C1-hydroxyl moiety of ED4 is then allowed to react with trichloroacetonitrile to give the TCA derivative ED5 suitable for coupling using diazabicycloundecane and dichloromethane, and mixing at room temperature for 2 hours. Monomer C, havinga free C4-hydroxyl group, was then coupled with the disaccharide ED5 in the presence of triethylsilyl triflate and mixed at −20° C. for 2 hours to give the trisaccharide EDC Trimer.
Synthesis of the EDCBA Pentamer
The EDCBA Pentamer was prepared using the following procedure:
The preparation of EDCBA Pentamer is accomplished in two parts as follows. In part 1, the EDC Trimer, a diacetate intermediate, is prepared for the coupling reaction with Dimer BA by initially opening the anhydro moiety and acetylation of the resulting hydroxyl groups to give the tetraacetate derivative EDC1. This occurs by reacting the EDC Trimer with boron trifluoride etherate, acetic anhydride and dichlormethane and stirring between −10° C. and room temperature for 3 hours. The C1-Acetate of EDC1 is then hydrolyzed to give the alcohol, EDC2, by reacting EDC1 with benzylamine [BnNH2] and tetrahydrofuran and mixing at −10° C. for 3 hours. The C1-hydroxyl of EDC2 is then reacted with trichloroacetonitrile and diazabicycloundecane, with mixing at room temperature for 2 hours, to give the trichloro acetamidate (TCA) derivative EDC3 suitable for coupling.
In Part 2 of the EDCBA Pentameter synthesis, the Dimer BA, having a free C4-hydroxyl group, is coupled with trisaccharide EDC3 in the presence of triethylsilyltriflate at −30° C. mixing for 2 hours to give the pentasaccharide EDCBA1. The levulinyl ester on C2 of sugar D in EDCBA1 is hydrolyzed with a mixture of deprotecting agents, hydrazine hydrate and hydrazine acetate and stiffing at room temperature for 3 hours to give the C2-hydroxyl containing intermediate EDCBA2. The C2-hydroxyl moiety on sugar D of EDCBA2 is then alkylated with dihydropyran (DHP) in the presence of camphor sulfonic acid (CSA) and tetrahydrofuran with mixing at room temperature for 3 hours to give the tetrahydropyranyl ether (THP) derivative, EDCBA Pentamer.
…………………………
A fast and effective hydrogenation process of protected pentasaccharide: A key step in the synthesis of fondaparinux sodium, Org Process Res Dev 2013, 17: 869, http://pubs.acs.org/doi/full/10.1021/op300367c
An improved method for the simultaneous removal of O-benzyl and N-carboxybenzyl groups as well as reducing azide groups to amines in protected heparin-like pentasaccharides, a key process in fondaparinux sodium synthesis, is reported. Under catalytic transfer hydrogenation conditions, using readily available and inexpensive ammonium formate, the hydrogenolysis is done in less than an hour in good yield and purity. This procedure represents a major advantage over the previously published procedures, the latter of which involve several hours/days of hydrogenation reaction under catalytic reduction using gaseous hydrogen.

Synthesis of Compound 1 (FONDAPARINUX)
Methyl O-(2-deoxy-6-O-sulfo-2-(sulfoamino)-α-d-glucopyranosyl)-(1→4)-O-(β-d-glucopyranuronosyl-(1→4)-O-2-deoxy-3,6-di-O-sulfo-2-(sulfamino)-α-d-glucopyranosyl-(1→4)-O-2-O-sulfo-α-L-iodpyranuronosyl-(1→4)-2-deoxy-6-O-sulfo-2-(sulfamino)-α-d-glucopyranoside) decasodium salt (1):
Pentasaccharide 5 (7.0 g, 4.9 mmol) was dissolved in water (140 mL) and pH was adjusted to 9.5 by the addition of solid Na2CO3. The sulfur trioxide–pyridine complex (11.8 g, 73.5 mmol) was slowly added, maintaining the pH of the reaction mixture at the range of 9.0–9.5 by dropwise addition of 5.0 M NaOH solution. When reaction was completed (HPLC), the mixture was concentrated under vacuum to 1/3 of volume and purified by Sephadex G-25 column (isocratic 0.2 M NaCl) and Dowex 50WX4 Na+ (gradient 0.2–2.0 M NaCl). Crude material 1was desalted using a Sephadex G-25 column (elution by water), and next the fractions that contained the product were treated with activated charcoal (150% of the weight of crude product) and stirred for several hours at 50 °C. Additional purification by ion-exchange column and desalting afforded product 1 (FONDAPARINUX .10 Na) in 50% yield (4.2g, 96% purity).
1H NMR (D2O) δ: 5.68 (d, J = 3.8 Hz, 1H, H-1A), 5.56 (d, J = 3.4 Hz, 1H, H-1C), 5.24 (d, J = 3.8 Hz, 1H, H-1D), 5.07 (d, J = 3.5 Hz, 1H, H-1E), 4.68 (d, J = 7.9 Hz, 1H, H-5D), 4.54 (dd, J = 11.4, 2.2 Hz, 1H, H-1B), 4.48–4.34 (m, 6H, H-6C, 6E, 6′E, 6A, 3B, 2D), 4.33–4.30 (m, 1H, H-6′C), 4.25–4.17 (m, 4H, H-4D, 3D, 6′A, 5C), 4.06–3.98 (m, 2H, H-4C, 5E), 3.94 (dd, J = 9.7, 2.2 Hz, 1H, H-5A), 3.92–3.86 (m, 2HH-3B, H-4B), 3.85–3.80 (m, 2H, H-5B, 4E), 3.73–3.60 (m, 3H, H-3E, 3A, 4A), 3.53–3.44 (m, 2H, H-2C, H-2B), 3.47 (s, 3H, OMe), 3.34 (dd, J = 10.2, 3.7 Hz, 1H, H-2E), 3.31 (dd, J = 10.2, 3.7 Hz, 1H, H-2A);
13C NMR (151 MHz, D2O) δ: 175.26, 174.08, 101.06, 99.47, 98.24, 97.47, 96.05, 77.03, 77.00, 76.75, 76.26, 76.19, 76.05, 75.95, 72.83, 72.71, 71.07, 70.17, 70.08, 69.75, 69.69, 69.51, 68.95, 68.48, 66.65, 66.27, 65.93, 57.88, 57.64, 56.57, 55.38,
MS: monoisotopic mass C31H43N3O49S85 calcd 1507.1, found:
ES(−) 752.6 [(M – 2H+)/2]−, 501.6 [(M – 3H+)/3]−, 474.8 [(M – SO3 – 2H+)]−, 376.5 [(M – 4H+)/4]−,
356.2 [(M – SO3 – 3H+)]−;
[α]D = 49.0 (c = 0.630, H2O)
1H NMR AND 13CNMR OF FONDAPARINUX. 10 Na
AT
………………
SYNTHESIS
In the synthesis of Fondaparinux sodium, the monomers XII, XVIII, XXVII, XXXVIII, XXXXI and dimers XIX, XX, XL described herein may be made either by processes described in the art or, by a process as described herein. The XII and XVIII monomers may then linked to form a disaccharide XX, XXXIX and XXVII monomers may then linked to form a disaccharide XL, XLIII and XX dimers may then linked to form a tetrasaccharide, XLVII tetramer and XLV monomer may be linked to form a pentasaccharide (XLVIII) pentamer. The XLVIII pentamer is an intermediate that may be converted through a series of reactions to fondaparinux sodium. This strategy described herein provides an efficient method for multi-kilogram preparation of fondaparinux in high yields and highly stereoselective purity.
Fondaparinux sodium (LIII) was prepared in 3 synthetic steps from O – S pentasaccharide (L) using the following procedure:
Fondaparinux Sodium (LIII)
Preparation of Fondaparinux sodium (LIII)—
N- sulfonation of Deprotected Pentasaccharide (LI) methyl 0-2-deoxy-3,6-di-0- sulfo-2-(sulfoamino)-a-D-glucopyranosyl-(l— >4)-0-2-0-sulfo-a-L- idopyranurosyl-( 1— >4)-2-deoxy-6-0-sulfo-2-(sulfoamino)-a-D-glucopyranoside,decasodium salt
A solution of deprotected pentasaccharide (LI) (145 gm) in water (2.54 V) was adjusted to a pH of 9.5 – 10.5 with 1 N NaOH solution. S03-pyridine complex (156 gm) was added into 3 lots every 15 min, the pH being maintained at 9.5-10.5 by automatic addition of 1 N NaOH. The mixture was stirred for 2 hrs at RT, during this aqueous NaOH (IN solution) was added to maintain pH at 9.5 – 10.5. After neutralization to pH 7 – 7.5 by addition of HC1 solution, the mixture was evaporated using vacuum. The residue was dissolved in water (1.6 L) at RT, to this solution was added acetone (1.6 L) at RT. The reaction mass was cooled to 5°C – 1 0 °C and stirred for 1 hr. The solid was filtered and washed with cold acetone: water (1 :1). The clear filtrate was distilled off completely under vacuum below 55°C. The residue was dissolved in water (1.6 L) at RT, and to this solution was added acetone(1.6 L) at RT. The mixture was cooled to 5 to 10°C and stirred for 1 hr. The solid was filtered and washed with cold acetone/water (1 :1). The clear filtrate was distilled off completely under vacuum below 55°C. The residue was dissolved in water (0.7 L) and charcoal (40 gm) was added at RT. The mixture was stirred for 30 min at RT then filtered. To the filtrate was added charcoal (40 gm) at RT. The mixture was stirred for 30 min at RT then filtered. To the filtrate was added charcoal (40 gm) at RT. The mixture was stirred for 30 min at RT then filtered. The pH of the clear filtrate was adjusted to 8.0 – 8.5 with IN NaOH solution and distilled off completely under vacuum below 55 °C. The residue was dissolved in 0.5 M NaCl solution and layered onto a column of Dowex® 1×2 -400 resins using a gradient of NaCl solution (0.5 to 10M). The product fractions were combined and distilled off under vacuum below 55 °C up to 1 – 2 L volume. The solid was filtered off and the clear filtrate was distilled off under vacuum below 55 °C up to slurry stage and subjected to azeotropic distillation with methanol two times. The solid residue was stirred with methanol (2.13 L) at RT for 1 hr and the solid was filtered off and washed with methanol. The wet solid was again stirred with methanol (2.13 L) at RT for 1 hr and the solid was filtered off and washed with methanol. The wet solid was again stirred with methanol (2.13 L) at RT for 1 hr and the solid was filtered off and washed with methanol. The above solid was dissolved in water and the pH adjusted to 4 – 4.5 with IN HC1 solution and charcoalized three times with 26 gm of charcoal at RT for 15-30 minutes and filtered off. To the clear filtrate was added 0.39 kg of NaCl, then methanol was added (35 volume) at RT and the mixture was stirred for 15-30 minutes. The solution was decanted and the sticky mass was stirred with methanol (0.65 L) at RT for 15-30 minutes. The solid was filtered off and dissolved in water, and the pH adjusted to 8 – 8.5 with IN NaOH solution. The solution was filtered through 0.45 micron paper & distilled off completely under vacuum below 55°C. The solution was subjected to azeotropic distillation with methanol to give highly pure fondaparinux sodium (97.17 gm) (HPLC purity 99.7%).
SOR Results
Three batches of product made in accordance with the present processes provided the following stereoisomeric optical rotation results:
Specification: Between +50.0° and +60.0°.
Batch- 1 = +55.1°
Batch-2 = +55.7° Batch-3 = +55.4°.
INTERMEDIATES
Synthetic Procedures
The following abbreviations are used herein: Ac is acetyl; MS is molecular sieve; DMF is dimethyl formamide; Bn is benzyl; MDC is dichloromethane; THF is tetrahydrofuran; TFA is trifluoro acetic acid; MeOH is methanol; RT is room temperature; Ac2O is acetic anhydride; HBr is hydrogen bromide; EtOAc is ethyl acetate; Cbz is benzyloxycarbonyl; CADS is chloro acetyl disaccharide; HDS is hydroxy disaccharide; NMP is N-methylpyrrolidone.
Methyl 3-O-benzyl-4-O-monochloro acetyl-β-L-idopyranuronate
Route of Synthesis for α-Methyl-6-o-acetyl-3-o-benzyl-2-(benzyloxy carbonyl)amino-2-deoxy-α-D-glucopyranoside
Methyl 6-O-acetyl-3-O-benzyl-2-(benzyloxy carbonyl)amino-2-deoxy-4-O-(methyl-2-O acetyl-3-O-benzyl-α-L-idopyranosyluronate)-glucopyranoside
Route of Synthesis for 1,6-Anhydro-2-azido-3-O-acetyl-2-deoxy-beta-D-glucopyranose
Route of synthesis for Methyl 2,3-di-O-benzyl-4-O-chloroacetyl-beta-D-glucopyranuronate
Route of synthesis for 3-O-Acetyl-1,6-anhydro-2-azido-4-O-2,3-di-O-benzyl-4-O-chloroacetyl-beta-D-glucopyranosyl methyluronate-beta-D-glucopyranose
(or)
3-O-Acetyl-1,6-anhydro-2-azido-2-deoxy-4-O-(methyl 2,3-di-O-benzyl-4-O-chloroacetyl-beta-D-glucopyranosyluronate)-beta-D-glucopyranose
Route of Synthesis for 1,6-Anhydro-2-azido-3,4-di-O-benzyl-2-deoxy-beta-D-glucopyranose
Synthesis of Disaccharide XLIII
Disaccharide XLIII was prepared in 2 synthetic steps from CADS sugar (XL) using the following procedure:
CADS sugar XL was acetylated at the anomeric carbon using AC2O and TFA to give acetyl derivative XLII. This step was carried out using the reactants CADS, AC2O and TFA, stirring in an ice water bath for about 5-24 hours, preferably 20 hours, and evaporating to residue under vacuum. Residue was recrystallized in ether. Acetyl CADS (XLII) was brominated at the anomeric carbon using titanium tetra bromide in MDC andethylacetate and stirring at 20° C.-50° C. for 6-16 hours, preferably 6 hours, to give the bromo derivative, (XLIII) after work-up and recrystallization from solvent/alcohol.
Synthesis of the Monosaccharide (XLV)
The monosaccharide (XLV) was prepared in 2 synthetic steps from monomer (XLI) using the following procedure:
Mono sugar (XLI) was acetylated at the anomeric carbon using AC2O and TFA to give acetyl derivative (XLIV). This step was carried out using the reactants Mono sugar (XLI), AC2O and TFA, stirring in an ice water bath for about 5-24 hours, preferably 24 hours, and evaporating to residue under vacuum. Residue was recrystallized in ether. Acetyl Mono sugar (XLIV) was brominated at the anomeric carbon using titanium tetra bromide in MDC and ethyl acetate and stirring at 20° C.-50° C. for 6-20 hours, preferably 16 hours, to give the bromo derivative, (XLV) after work-up and recrystallization from ether.
Synthesis of the Hydroxy Tetrasaccharide (XLVII)
The hydroxy tetrasaccharide (XLVII) was prepared in 2 synthetic steps from disaccharide (XLIII) and HDS (XX) using the following procedure:
Disaccharide (XLIII), was coupled with disaccharide (XX) in the presence of silver carbonate, silver per chlorate and 4 A° MS in MDC and stirred at ambient temperature for 5-12 hrs, preferably 4-6 hours, in the dark followed by work-up and purification in water/methanol to give the tetrasaccharide (XLVI). The d echloroacetylation of tetrasaccharide (XLVI) was carried out in THF, ethanol and pyridine in the presence of thiourea at reflux for 6 to 20 hrs, preferably 12 hours, to give the hydroxy tetrasaccharide (XLVIII).
Synthesis of the Pentasaccharide (XLVIII)
The pentasaccharide (XLVIII) was prepared in 2 synthetic steps from monosaccharide (XLV) and tetrasaccharide (XLVII) using the following procedure:
Monosaccharide (XLV), was coupled with tetrasaccharide (XLVII) in the presence of 2,4,6-collidine, silver triflate and 4 A° MS in MDC and stirred at −10° C. to −20° C. for 1 hr in the dark followed by work-up and purification by column chromatography to give the pentasaccharide (XLVIII).
Synthesis of OS Pentasaccharide (L)
The OS pentasaccharide (L) was prepared in 2 synthetic steps from pentasaccharide (XLVIII) using the following procedure:
Pentasaccharide (XLVIII) was deacetylated in the presence of NaOH in mixture of solvents of MDC, methanol and water at 0° C. to 35° C., for 1-2 hrs followed by work-up and distillation to obtain deacetylated pentasaccharide (XLIX) which was subjected to O-sulfonation in DMF in the presence of SO3-trimethylamine (TMA) at 50° C. to 100° C., preferably 50° C.-55° C., for 6-24 hrs, preferably 12 hours, followed by salt removal through Sephadex® resin and column chromatography purification, then pH adjustment by dilute NaOH to give OS pentasaccharide (L).
…………………………
INTERMEDIATE
highly pure 4-Ο-β-ϋ- glucopyranosyl- 1 ,6-anhydro- -D-glucopyranose
FORMULA II
Example 1 : Preparation and purification of 4-0- -D-grucopyranosyl-L6-anhvdro- -D- glucopyranose
A solution of pentachlorophenyl 2,3,6,2′,3′,4′,6′-hepta-(9-acetyl- -D-ceilobioside represented by Formula I;

(400 g) in isopropyl alcohol (4 L) at ambient temperature was cooled to 2°C to 5°C and pulverized potassium hydroxide (355 g) was added to it. This reaction mixture was stirred and the temperature was allowed to rise to ambient temperature. At ambient temperature, the mixture was stirred until the reaction was complete (about 2 hours). The mixture was then heated to 50°C to 55°C and stirred for 30 minutes. The solid obtained was filtered and washed with isopropyl alcohol (400 mL). The solid was stirred with isopropyl alcohol (2.8 L) at 50°C for 30 minutes followed by filtering and washing with isopropyl alcohol (400 mL). The resultant solid was suspended into methanol (800 mL to 1600 mL) followed by cooling to 2°C to 5°C. The pH of the suspension was adjusted to 2 to 3 using 15% methanolic hydrochloride. The solid so obtained was filtered and washed with methanol (400 mL). Solvent was recovered from the filtrate to dryness under vacuum to obtain the pure compound of Formula II as foamy solid.
Yield: 142 g
Example 2: Preparation and purification of 4-Q- -D-grucopyranosyl-l,6-anhvdro- -D- glucopyranose
A solution of pentachlorophenyl 2,3,6,2 ,3 ^ ^‘-hepta-O-acetyl- -D-cellobioside of Formula I (100 g) in methanol (300 mL) at ambient temperature was cooled to 2°C to 5°C and pulverized potassium hydroxide (88.6 g) was added to it. This reaction mixture was stirred and the temperature was allowed to rise to ambient temperature. At ambient temperature, the mixture was stirred until the reaction was complete (about 2 hours). The mixture was cooled to 2°C to 5°C and 15% methanolic hydrogen chloride was added to it until the pH of the mixture reached 2 to 3. At this pH, the reaction mixture was filtered and the residual solid was washed with methanol (100 mL). The solvent was recovered from the filtrate under vacuum. The solid material so obtained was stirred with dichloromethane (500 mL) followed by removal of solvent through decantation/filtration. The resultant solid was stirred with isopropyl alcohol (500 mL), filtered and dried to obtain the pure compound of Formula II.
Yield: 29 g
………………………
SYNTHESIS
Synthesis of Fondaparinux
Fondaparinux was prepared using the following procedure:
Conversion of FPP (also referred to a Fully Protected Pentamer) to FondaparinuxSodium:
Reagents: 1. NaOH, H202, LiOH, Dioxane, RT, 24-48 h; 2. Py.S03, DMF, 60°C, 2h, CG-161 purification; 3. 10% Pd/C, H2, 72h; 4. (a) Py.S03, NaOH, NH4OAc, 12h, (b) HiQ NH4OAc/ NaCl ion-exchange, Sephadex Desalt and (c) HiQ NaCl ion-exchange, Sephadex Desalt. The ester moieties in EDCBA Pentamer-CB were hydrolyzed with sodium and lithium hydroxide in the presence of hydrogen peroxide in dioxane mixing at room temperature for 24- 48 hours to give the pentasaccharide intermediate API1-CB. The five hydroxyl moieties in API1-CB were sulfated using a pyridine-sulfur trioxide complex in dimethylformamide, mixing at 60°C for 2 hours and then purified using column chromatography (CG-161), to give the pentasulfated pentasaccharide API2-CB. The intermediate API2-CB was then hydrogenated to reduce the three azides on sugars E, C and A to amines and the reductive deprotection of the six benzyl ethers to their corresponding hydroxyl groups to form the intermediate API3-CB. This transformation occurs by reacting API2-CB with 10% palladium/carbon catalyst with hydrogen gas for 72 hours. The three amines on API3-CB were then sulfated using the pyridine-sulfur trioxide complex in sodium hydroxide and ammonium acetate, allowing the reaction to proceed for 12 hours . The crude fondaparinux is purified and is subsequently converted to its salt form. The crude mixture was purified using an ion-exchange chromatographic column (HiQ resin) followed by desalting using a size exclusion resin or gel filtration (Biorad Sephadex G25) to give the final product, fondaparinux sodium.
Preparation of Fondaparinux Sodium – Step 4: N-Sulfation of API-3-CB:
Methyl 0-2-deoxy-6-0-sulfo-2-(sulfoamino)-a-D-glucopyranosyl-(l→4)-0^-D- glucopyranuronosyl-(l→4)-0-2-deoxy-3,6-di-0-sulfo-2-(sulfoamino)-a-D-glucopyranosyl- (l→4)-0-2-0-sulfo-a-L-idopyranuronosyl-(l→4)-2-deoxy-6-0-sulfo-2-(sulfoamino)-a-D- glucopyranoside, decasodium salt
To a solution of 25.4 gram (16.80 mmol, leq) of API-3-CB in 847 mL of water was slowly added 66.85 gram (446.88 mmol, 25eq) of sulfur trioxide-pyridine complex, maintaining the pH of the reaction mixture at pH 9-9.5 during the addition using 2N sodium hydroxide solution. The reaction was allowed to stir for 4 hours at pH 9.0 – 9.5. When reaction was completed, the pH was adjusted 7.0 by using 70 mL of 50 mmol Ammonium acetate solution pH -3.5. The resulting N-Sulfated Cellobiose mixture was purified using Ion-Exchange
Chromatographic Column followed by desalting using size exclusion resin to gave gram ( %) of the purified Fondaparinux Sodium form.
To a solution of 942 g (0.63 mol) of API3 in 46 L of water was slowly added 3.25 Kg (20.4 mol, 32 eq) of Sulfur trioxide-pyridine complex, maintaining the pH of the reaction mixture at pH 9-9.5 during the addition using 2 N sodium hydroxide solution. The reaction was allowed to stir for 4-6 hours at pH 9.0-9.5. When reaction was complete, the pH was adjusted to pH 7.0 using 50 mM solution of Ammonium acetate at pH 3.5. The resulting N- sulfated EDCBA(OS03)5(NHS03)3 mixture was purified using Ion-Exchange Chromatographic Column (Varian Preparative 15 cm HiQ Column) followed by desalting using a size exclusion resin or gel filtration (Biorad G25). The resulting mixture was then treated with activated charcoal and the purification by ion-exchange and desalting were repeated to give 516 g (47.6% yield) of the purified Fondaparinux sodium form.
INT
SCHEME 1 – Synthesis of Monomer A-2 & AMod5 fBuildinq Block Al
Reagents: 1. NaOMe, MeOH, RT, 2hr, 50wx resin; 2. (Bu3Sn)20 (0.8equiv), ACN, MS, reflux, 3h; 3.l2 (1.5 equiv), 5°C to RT, 2h; 4. NaH (2 equiv), DMF, p-MeOC6H4CH2Br (PMB-Br, 2.5 equiv), -20°C to RT, 2h; 5. NaN3, DMF, 120°C, 12h; 6. NaH, DMF, BnBr, 0°C to RT, 3h.; 7. BF3.Et20, Ac20, DCM, -20°C to RT, 3h; 8. (a) TMS-I, TBAI, RT, 2h; (b) DIPEA, MeOH, 16h, RT; 9. NaOMe, Dowex 50WX8-100 resin H+ form, RT, 3h; 10. Pyridine, Bz-CI, -40°C to -10°C, 2h;
Scheme 2 – Synthesis of Monomer B-1 and BMod6 fBuildinq Block B1
Reagents: 1. NaH, BnBr, THF, DMF, 0° to 65°C, 3h; 2. 66% Acetic Acid/H20, 40 °C, 16h; 3. Nal04, (Bu)4NBr, DCM, H20, Dark, 3h; 4. (PhS)3CH, n-BuLi, THF, -78 °C, 3h; 5. CuCI2/CuO, MeOH, H20, 3h; 6. 90% TFA/H20, DCM, RT, 2h; 7. DMF, CSA 2-methoxypropene, 0° to RT, 16hrs; MeOH, TEA. 8. Lev20, DIPEA, RT, 16h; 9. 90% TFA, RT, 4h; 10. Imidazole, TBDPSi-CI, RT, 3h; 11. Pyridine, BzCI, RT, 3h; 12. TBAF, RT, 3h; 13. TCA, DBU, RT, 2h; Also see, e.g., Bonnaffe et al., Tetrahedron Lett., 41, 307-311, 2000; Bonnaffe et al., Carbohydr. Res., 2003, 338, 681-686, 2003; and Seeberger et al., J. Org. Chem., 2003, 68, 7559- 7561, 2003.
……………………..
Carbohydrate Research, 2012 , vol. 361, p. 155 – 161
1H NMR (D2O) δ: 5.68 (d, J = 3.8 Hz, 1H, H-1D), 5.56 (d, J = 3.4 Hz, 1H, H-1F), 5.24 (d, J = 3.8 Hz, 1H, H-1G), 5.07 (d, J = 3.5 Hz, 1H, H-1H), 4.68 (d, J = 7.9 Hz, 1H, H-5G), 4.54 (dd, J = 11.4, 2.2 Hz, 1H, H-1E), 4.48-4.34 (m, 6H, H-6F, 6H, 6′H, 6D, 3E, 2G), 4.33-4.30 (m, 1H, H-6′F), 4.25-4.17 (m, 4H, H-4G, 3G, 6′D, 5F), 4.06-3.98 (m, 2H, H-4F, 5H), 3.94 (dd, J = 9.7, 2.2 Hz, 1H, H-5D), 3.92-3.86 (m, 2H,H-3E, H-4E), 3.85-3.80 (m, 2H, H-5E, 4H), 3.73-3.60 (m, 3H, H-3H, 3D, 4D), 3.53-3.44 (m, 2H, H-2F, H-2E), 3.47 (s, 3H, OMe), 3.34 (dd, J = 10.2, 3.7 Hz, 1H, H-2H), 3.31(dd, J = 10.2, 3.7 Hz, 1H, H-2D)
FONDAPARINUX
……………………………………..
Synthesis of intermediates
Synthetic Procedures
The following abbreviations are used herein: Ac is acetyl; ACN is acetonitrile; MS is molecular sieves; DMF is dimethyl formamide; PMB is p-methoxybenzyl; Bn is benzyl; DCM is dichloromethane; THF is tetrahydrofuran; TFA is trifluoro acetic acid; CSA is camphor sulfonic acid; TEA is triethylamine; MeOH is methanol; DMAP is dimethylaminopyridine; RT is room temperature; CAN is ceric ammonium nitrate; Ac2O is acetic anhydride; HBr is hydrogen bromide; TEMPO is tetramethylpiperidine-N-oxide; TBACl is tetrabutyl ammonium chloride; EtOAc is ethyl acetate; HOBT is hydroxybenzotriazole; DCC is dicyclohexylcarbodiimide; Lev is levunlinyl; TBDPS is tertiary-butyl diphenylsilyl; TCA is trichloroacetonitrile; O-TCA is O-trichloroacetimidate; Lev2O is levulinic anhydride; DIPEA is diisopropylethylamine; Bz is benzoyl; TBAF is tetrabutylammonium fluoride; DBU is diazabicycloundecane; BF3.Et2O is boron trifluoride etherate; TMSI is trimethylsilyl iodide; TBAI is tetrabutylammonium iodide; TES-Tf is triethylsilyl trifluoromethanesulfonate (triethylsilyl triflate); DHP is dihydropyran; PTS is p-toluenesulfonic acid.
The monomers used in the processes described herein may be prepared as described in the art, or can be prepared using the methods described herein.
The synthesis of Monomer A-2 (CAS Registry Number 134221-42-4) has been described in the following references: Arndt et al., Organic Letters, 5(22), 4179-4182, 2003; Sakairi et al., Bulletin of the Chemical Society of Japan, 67(6), 1756-8, 1994; and Sakairi et al., Journal of the Chemical Society, Chemical Communications, (5), 289-90, 1991, and the references cited therein, which are hereby incorporated by reference in their entireties.
Monomer C(CAS Registry Number 87326-68-9) can be synthesized using the methods described in the following references: Ganguli et al., Tetrahedron: Asymmetry, 16(2), 411-424, 2005; Izumi et al., Journal of Organic Chemistry, 62(4), 992-998, 1997; Van Boeckel et al., Recueil: Journal of the Royal Netherlands Chemical Society, 102(9), 415-16, 1983; Wessel et al.,Helvetica Chimica Acta, 72(6), 1268-77, 1989; Petitou et al., U.S. Pat. No. 4,818,816 and references cited therein, which are hereby incorporated by reference in their entireties.
Monomer E (CAS Registry Number 55682-48-9) can be synthesized using the methods described in the following literature references: Hawley et al., European Journal of Organic Chemistry, (12), 1925-1936, 2002; Dondoni et al., Journal of Organic Chemistry, 67(13), 4475-4486, 2002; Van der Klein et al., Tetrahedron, 48(22), 4649-58, 1992; Hori et al., Journal of Organic Chemistry, 54(6), 1346-53, 1989; Sakairi et al., Bulletin of the Chemical Society of Japan, 67(6), 1756-8, 1994; Tailler et al.,Journal of the Chemical Society, Perkin Transactions 1: Organic and Bio-Organic Chemistry, (23), 3163-4, (1972-1999) (1992); Paulsen et al., Chemische Berichte, 111(6), 2334-47, 1978; Dasgupta et al., Synthesis, (8), 626-8, 1988; Paulsen et al., Angewandte Chemie, 87(15), 547-8, 1975; and references cited therein, which are hereby incorporated by reference in their entireties.
Monomer B-1 (CAS Registry Number 444118-44-9) can be synthesized using the methods described in the following literature references: Lohman et al., Journal of Organic Chemistry, 68(19), 7559-7561, 2003; Orgueira et al., Chemistry—A European Journal, 9(1), 140-169, 2003; Manabe et al., Journal of the American Chemical Society, 128(33), 10666-10667, 2006; Orgueira et al., Angewandte Chemie, International Edition, 41(12), 2128-2131, 2002; and references cited therein, which are hereby incorporated by reference in their entireties.
Synthesis of Monomer D
Monomer D was prepared in 8 synthetic steps from glucose pentaacetate using the following procedure:
Synthesis of Monomer D
Monomer D was prepared in 8 synthetic steps from glucose pentaacetate using the following procedure:
Pentaacetate SM-B was brominated at the anomeric carbon using HBr in acetic acid to give bromide derivative IntD1. This step was carried out using the reactants SM-B, 33% hydrogen bromide, acetic acid and dichloromethane, stirring in an ice water bath for about 3 hours and evaporating at room temperature. IntD1 was reductively cyclized with sodium borohydride and tetrabutylammonium iodide in acetonitrile using 3 Å molecular sieves as dehydrating agent and stirring at 40° C. for 16 hours to give the acetal derivative, IntD2. The three acetyl groups in IntD2 were hydrolyzed by heating with sodium methoxide in methanol at 50° C. for 3 hours and the reaction mixture was neutralized using Dowex 50WX8-100 resin (Aldrich) in the acid form to give the trihydroxy acetal derivative IntD3.
The C4 and C6 hydroxyls of IntD3 were protected by mixing with benzaldehyde dimethyl acetate and camphor sulphonic acid at 50° C. for 2 hours to give the benzylidene-acetal derivative IntD4. The free hydroxyl at the C3 position of IntD4 was deprotonated with sodium hydride in THF as solvent at 0° C. and alkylated with benzyl bromide in THF, and allowing the reaction mixture to warm to room temperature with stirring to give the benzyl ether IntD5. The benzylidene moiety of IntD5 was deprotected by adding trifluoroacetic acid in dichloromethane at 0° C. and allowing it to warm to room temperature for 16 hours to give IntD6 with a primary hydroxyl group. IntD6 was then oxidized with TEMPO (2,2,6,6-tetramethyl-1-piperidine-N-oxide) in the presence of tetrabutylammonium chloride, sodium bromide, ethyl acetate, sodium chlorate and sodium bicarbonate, with stirring at room temperature for 16 hours to form the carboxylic acid derivative IntD7. The acid IntD7 was esterified with benzyl alcohol and dicyclohexylcarbodiimide (other reactants being hydroxybenzotriazole and triethylamine) with stirring at room temperature for 16 hours to give Monomer D.
Synthesis of the BA Dimer
The BA Dimer was prepared in 12 synthetic steps from Monomer B1 and Monomer A2 using the following procedure:
The C4-hydroxyl of Monomer B-1 was levulinated using levulinic anhydride and diisopropylethylamine (DIPEA) with mixing at room temperature for 16 hours to give the levulinate ester BMod1, which was followed by hydrolysis of the acetonide with 90% trifluoroacetic acid and mixing at room temperature for 4 hours to give the diol BMod2. The C1 hydroxyl of the diol BMod2 was silylated with tert-butyldiphenylsilylchloride by mixing at room temperature for 3 hours to give silyl derivative BMod3. The C2-hydroxyl was then benzoylated with benzoyl chloride in pyridine, and mixed at room temperature for 3 hours to give compound BMod4. The silyl group on BMod4 was then deprotected with tert-butyl ammonium fluoride and mixing at room temperature for 3 hours to give the C1-hydroyl BMod5. The C1-hydroxyl is then allowed to react with trichloroacetonitrile in the presence of diazobicycloundecane (DBU) and mixing at room temperature for 2 hours to give the trichloroacetamidate (TCA) derivative BMod6, which suitable for coupling, for example with Monomer A-2.
Monomer A-2 was prepared for coupling by opening the anhydro moiety with BF3.Et2O followed by acetylation of the resulting hydroxyl groups to give the triacetate derivative AMod1.
Monomer A2 was prepared for the coupling reaction by opening the anhydro moiety and acetylation of the resulting hydroxyl groups to give the triacetate derivative AMod1. This transformation occurs using boron trifluoride etherate, acetic anhydride and dichloromethane, between −20° C. and room temperature for 3 hours. The C1-Acetate of AMod1 was then hydrolyzed and methylated in two steps to give the diacetate AMod3. That is, first AMod1 was reacted with trimethylsilyl iodide and mixed at room temperature for 2 hours, then reacted with and tetrabutyl ammonium iodide. This mixture was reacted with diisoproylethylamine and methanol and stirred for 16 hours at room temperature, thus forming AMod3. The C4 and C6 acetates of AMod3 are hydrolyzed with sodium methoxide to give the diol Amod4. The AMod3 mixture was also subjected to mixing at room temperature for 3 hours with Dowex 50 Wx4x8-100 resin in the acid form for neutralization. This formed Amod4. The C6-hydroxyl of AMod4 is then benzoylated by treating with benzoyl chloride in pyridine at −40° C. and then allowing it to warm up to −10° C. over 2 hours to give AMod5.
Coupling of monomer AMod5 with the free C4-hydroxyl group of BMod6 was performed in the presence of BF3.Et2O and dichloromethane with mixing between −20° C. and room temperature for 3 hours to provide disaccharide BA1. The C4-levulinyl moiety of the disaccharide was then hydrolyzed with hydrazine to give the BA Dimer, which is suitable for subsequent coupling reactions.
Synthesis of EDC Trimer
The EDC Trimer was prepared in 10 synthetic steps from Monomer E, Monomer D and Monomer C using the following procedure:
Monomer E was prepared for coupling by opening the anhydro moiety with BF3.Et2O followed by acetylation of the resulting hydroxyl groups to give diacetate EMod1. This occurs by the addition of Monomer E with boron trifluoride etherate, acetic anhydride and dichloromethane at −10° C., and allowing the reaction to warm to room temperature with stirring for 3 hours. The C1-Acetate of EMod1 is then hydrolyzed to give the alcohol, EMod2. This occurs by reacting Emod1 with hydrazine acetate and dimethylformamide and mixing at room temperature for 3 hours. The C1-hydroxyl of Emod2 is then reacted with trichloroacetonitrile to give the trichloro acetamidate (TCA) derivative EMod3 suitable for coupling, which reaction also employs diazabicycloundecane and dichloromethane and mixing at room temperature for 2 hours.
Monomer D, having a free C4-hydroxyl group, was coupled with monomer EMod3 in the presence of triethylsilyl triflate with mixing at −40° C. for 2 hours to give the disaccharide ED Dimer. The acetal on ring sugar D of the ED Dimer is hydrolyzed to give the C1,C2-diol ED1. This occurs by reacting the ED Dimer with 90% trifluoro acetic acid and mixing at room temperature for 4 hours. The C1-hydroxyl moiety of ED1 was then silylated with tert-butyldiphenylsilyl chloride to give the silyl derivative ED2. The C2-hydroxyl of ED2 was then allowed to react with levulinic anhydride in the presence of dimethylaminopyridine (DMAP) and diethylisopropylamine for approximately 16 hours to give the levulinate ester ED3. The TBDPS moiety is then deprotected by removal with tert-butylammonium fluoride in acetic acid with mixing at room temperature for 3 hours to give ED4 having a C1-hydroxyl. The C1-hydroxyl moiety of ED4 was then allowed to react with trichloroacetonitrile to give the TCA derivative ED5, which is suitable for coupling.
The C1-hydroxyl moiety of ED4 is then allowed to react with trichloroacetonitrile to give the TCA derivative ED5 suitable for coupling using diazabicycloundecane and dichloromethane, and mixing at room temperature for 2 hours. Monomer C, having a free C4-hydroxyl group, was then coupled with the disaccharide ED5 in the presence of triethylsilyl triflate and mixed at −20° C. for 2 hours to give the trisaccharide EDC Trimer.
Synthesis of the EDCBA Pentamer
The EDCBA Pentamer was prepared using the following procedure:
The preparation of EDCBA Pentamer is accomplished in two parts as follows. In part 1, the EDC Trimer, a diacetate intermediate, is prepared for the coupling reaction with Dimer BA by initially opening the anhydro moiety and acetylation of the resulting hydroxyl groups to give the tetraacetate derivative EDC1. This occurs by reacting the EDC Trimer with boron trifluoride etherate, acetic anhydride and dichlormethane and stirring between −10° C. and room temperature for 3 hours. The C1-Acetate of EDC1 is then hydrolyzed to give the alcohol, EDC2, by reacting EDC1 with benzylamine [BnNH2] and tetrahydrofuran and mixing at −10° C. for 3 hours. The C1-hydroxyl of EDC2 is then reacted with trichloroacetonitrile and diazabicycloundecane, with mixing at room temperature for 2 hours, to give the trichloro acetamidate (TCA) derivative EDC3 suitable for coupling.
In Part 2 of the EDCBA Pentameter synthesis, the Dimer BA, having a free C4-hydroxyl group, is coupled with trisaccharide EDC3 in the presence of triethylsilyltriflate at −30° C. mixing for 2 hours to give the pentasaccharide EDCBA1. The levulinyl ester on C2 of sugar D in EDCBA1 is hydrolyzed with a mixture of deprotecting agents, hydrazine hydrate and hydrazine acetate and stiffing at room temperature for 3 hours to give the C2-hydroxyl containing intermediate EDCBA2. The C2-hydroxyl moiety on sugar D of EDCBA2 is then alkylated with dihydropyran (DHP) in the presence of camphor sulfonic acid (CSA) and tetrahydrofuran with mixing at room temperature for 3 hours to give the tetrahydropyranyl ether (THP) derivative, EDCBA Pentamer.
………………………………
Intermediates listed on the internet
Fondaparinux sodium Intermediates
Fondaparinux sodium N-4

……………………………….
Fondaparinux sodium N-3
114903-05-8

a-D-Glucopyranoside, Methyl O-2-azido-2-deoxy-3,4-bis-O-(phenylMethyl)-a-D-glucopyranosyl-(14) -O-2,3-bis-O-(phenylMethyl)-b-D-glucopyranuronosyl-(14)-O-2-azido- 2-deoxy-a-D-glucopyranosyl-(14)-O-3-O-(phenylMethyl)-a-L-idopyranu ronosyl-(14)-2-deoxy-2
FSC

114903-05-8

| |||||||||
| Description | |||||||||
| |||||||||
 | |||||||||
| |||||||||
| Description | |||||||||
| |||||||||
 | |||||||||
| |||||||||
| Description | |||||||||
| |||||||||
 | |||||||||
| |||||||||
| Description | |||||||||
| |||||||||
 | |||||||
| |||||||
| Description | |||||||
| |||||||
 | |||||||||
| |||||||||
| Description | |||||||||
| |||||||||
 | |||||||||
| |||||||||
| Description | |||||||||
| |||||||||
References
- “Medscape.com”. Retrieved 2009-01-23.
- “NEJM — Comparison of Fondaparinux and Enoxaparin in Acute Coronary Syndromes”. Retrieved 2009-01-23.
- Peters RJ, Joyner C, Bassand JP, et al. (February 2008). “The role of fondaparinux as an adjunct to thrombolytic therapy in acute myocardial infarction: a subgroup analysis of the OASIS-6 trial”.Eur. Heart J. 29 (3): 324–31. doi:10.1093/eurheartj/ehm616. PMID 18245119.
- WO 2013003001
- Synthesis of heparin fragments: A methyl alpha-pentaoside with high affinity for antithrombin III
Carbohydr Res 1987, 167: 67 - A fast and effective hydrogenation process of protected pentasaccharide: A key step in the synthesis of fondaparinux sodiumOrg Process Res Dev 2013, 17: 869, http://pubs.acs.org/doi/full/10.1021/op300367c
- WO 2012047174
- US 2012116066
- WO 2013011460 RANBAXY
- WO 2013115817
- The unique antithrombin III binding domain of heparin: A lead to new synthetic antithrombotics
Angew Chem Int Ed Engl 1993, 32(12): 1671 - Bioorganic and Medicinal Chemistry Letters, 1(2), p. 95-98 (1991).
- Carbohydrate Research, 101, p. 148-151 (1982),
- Chemistry – A European Journal, 2012 , vol. 18, 34 pg. 10643 – 10652
- Carbohydrate Research, 2012 , vol. 361, p. 155 – 161
- Analytical Chemistry, 2006 , vol. 78, 6 pg. 1774 – 1779
PATENTS
| US4818816 * | Oct 26, 1987 | Apr 4, 1989 | Choay, S.A. | Process for the organic synthesis of oligosaccharides and derivatives thereof |
| US6376663 * | Nov 29, 1996 | Apr 23, 2002 | Macquarie Research Ltd. | Desalting and purification of oligosaccharides and their derivatives |
| US7541445 * | Sep 6, 2002 | Jun 2, 2009 | Alchemia Limited | Synthetic heparin pentasaccharides |
| US20040048785 * | Jun 18, 2003 | Mar 11, 2004 | Societe L’oreal S.A. | C-glycoside compounds for stimulating the synthesis of glycosaminoglycans |
| US20040149200 * | Jun 11, 2002 | Aug 5, 2004 | Tsuyoshi Shimose | Crystals of an oligosaccharides and process for preparation thereof |
| US20110105418 * | Jul 30, 2010 | May 5, 2011 | Reliable Biopharmaceutical Corporation | Process for preparing fondaparinux sodium and intermediates useful in the synthesis thereof |
| WO2011014793A2 * | Jul 30, 2010 | Feb 3, 2011 | Reliable Biopharmaceutical Corporation | Process for preparing fondaparinux sodium and intermediates useful in the synthesis thereof | |
| AU2008200616A1 | Title not available | ||||
| JPS63218691A * | Title not available | ||||
| US4818816 | Oct 26, 1987 | Apr 4, 1989 | Choay, S.A. | Process for the organic synthesis of oligosaccharides and derivatives thereof | |
| US7468358 | Oct 27, 2004 | Dec 23, 2008 | Paringenix, Inc. | Method and medicament for sulfated polysaccharide treatment of heparin-induced thrombocytopenia (HIT) syndrome | |
| US84771910 | Title not available | ||||
| USPP23055709 | Title not available |
FONDAPARINUX



The three specialties available in the United States – dalteparin (Fragmin, Pfizer), enoxaparin (Lovenox, Sanofi-Aventis) and tinzaparin (Innohep, Bristol-Myers Squibb) – the first two are found in Brazil, enoxaparin under the names Lovenox, Cutenox and Dripanina.


3.

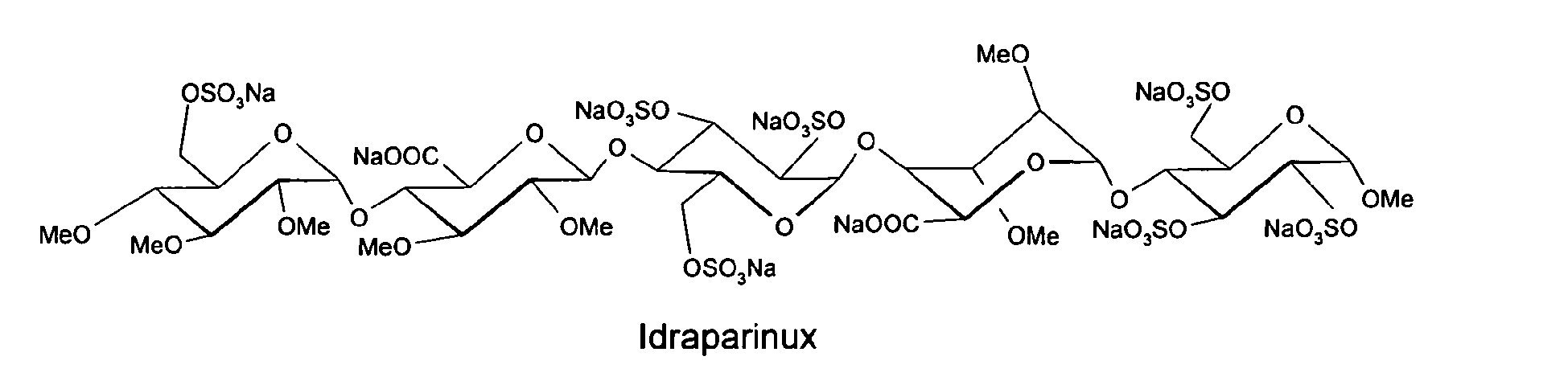







































































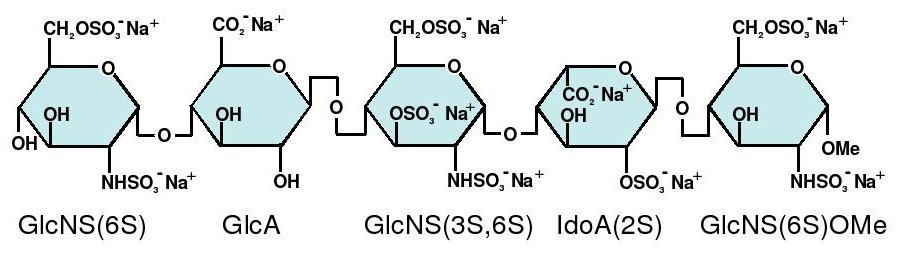
Hi i am Belvin Kushie from US I have just experience the wonders of Dr. Ogudugu love spell, that have been spread on the internet and worldwide, How he marvelously helped people all over the world to restored back their marriage life and get back lost lovers, and also help to win lottery. I contacted him after going through so many testimonies from different people how he help to bring back ex lover back, i told him about my husband that abandoned me about 8 months ago, and left home with all i had.. Dr. Ogudugu only told me to smile and have a rest of mind he will handle all in just 24 hours, After the second day my husband called me, i was just so shocked, i pick the call and couldn't believe my ears, he was really begging me to forgive him and making promises on phone.. He came back home and also got me a new car just for him to proof his love for me. i was so happy and called Dr.Ogudugu and thanked him, he only told me to share the good news all over the world ..Well if your need an effective and real spell caster contact Dr Ogudugu Via email: greatogudugu@gmail.com
ReplyDelete