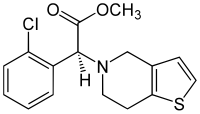
Clopidogrel (INN) is an oral, thienopyridine-class antiplatelet agent used to inhibit blood clots in coronary artery disease, peripheral vascular disease, and cerebrovascular disease. It is marketed by Bristol-Myers Squibb and Sanofi under the trade name Plavix. The drug works by irreversibly inhibiting a receptor called P2Y12, an adenosine diphosphate (ADP) chemoreceptor on platelet cell membranes. Adverse effects include hemorrhage, severe neutropenia, and thrombotic thrombocytopenic purpura.
Before the expiry of its patent, clopidogrel was the second-most prescribed drug in the world. In 2010, it grossed over US$9 billion in global sales.[1]
Medical use
Clopidogrel is used to prevent myocardial infarction (heart attack) and stroke in people who are at high risk in certain people at elevated risk of these events, including those with unstable angina, myocardial infarction (heart attack) with or without ST elevation, and those with established peripheral artery disease, recent heart attack, or ischemic stroke.
Treatment with clopidogrel or a related drug is recommended by the American Heart Association and the American College of Cardiology for people who
- Present for treatment with a heart attack with ST-elevation,[2] including
- A loading dose given in advance of percutaneous coronary intervention, followed by a full year of treatment for those receiving a vascular stent
- A loading dose given in advance of fibrinolytic therapy, continued for at least 14 days
- Present for treatment of a heart attack or unstable angina without ST-elevation .[3]
- Including a loading dose and maintenance therapy in those receiving PCI and unable to tolerate aspirin therapy
- Maintenance therapy for up to 12 months in those at medium to high risk for which a non-invasive treatment strategy is chosen
- In those with stable ischemic heart disease,[4] treatment with clopidogrel is described a a "reasonable" option for monotherapy in those who cannot tolerate aspirin, as is treatment with clopidogrel in combination with aspirin in certain high risk patients.
It is also used, along with aspirin (ASA), for the prevention of thrombosis after placement of an intracoronary stent[5] or as an alternative antiplatelet drug for people intolerant to aspirin.[6]
Clopidogrel's benefit is primarily in those who smoke cigarettes, with only slight benefit in those who do not.[7]
International guidelines granted the highest grade of recommendation for NSTE-ACS, PCI and stent,[] for clopidogrel in addition to ASA. Consensus-based therapeutic guidelines also recommend the use of clopidogrel rather than ASA for antiplatelet therapy in people with a history of gastric ulceration, as inhibition of the synthesis of prostaglandins by ASA can exacerbate this condition. In people with healed ASA-induced ulcers, however, those receiving ASA plus the proton pump inhibitor esomeprazole had a lower incidence of recurrent ulcer bleeding than those receiving clopidogrel.[8] However, prophylaxis with proton pump inhibitors along with clopidogrel following acute coronary syndrome may increase adverse cardiac outcomes, possibly due to inhibition of CYP2C19, which is required for the conversion of clopidogrel to its active form.[9][10][11] The European Medicines Agency has issued a public statement on a possible interaction between clopidogrel and proton pump inhibitors.[12] However, several cardiologists have voiced concern that the studies on which these warnings are based have many limitations and that it is not certain whether an interaction between clopidogrel and proton pump inhibitors is real.[13]
Adverse effects
Serious adverse drug reactions associated with clopidogrel therapy include:
- Severe neutropenia (low white blood cells) (incidence: one in 2,000)
- Thrombotic thrombocytopenic purpura (incidence: four per million patients treated)[14][15]
- Hemorrhage - the annual incidence of hemorrhage may be increased by the coadministration of aspirin.[16]
- Gastrointestinal hemorrhage (incidence: 2.0% annually)
- Cerebral hemorrhage (incidence: 0.1 to 0.4% annually)
- Use of nonsteroidal anti-inflammatory drugs is discouraged in those taking clopidogrel due to increased risk of digestive tract hemorrhage
- Bleeding in the postoperative period is especially problematic for patients after heart surgery, where clopidogrel is associated with a more than double the take-back for bleeding rate, as well as other complications. The take-back for bleeding occurs when chest tube clogging occurs in the setting of ongoing bleeding in early postoperative period. Often, if chest tube clogging can be avoided, and the chest tubes drain, the patient can be given platelets until the platelet defect is corrected and the bleeding ceases. But, if the bleeding continues, and the chest tubes occlude, then the patient will become hemodynamically unstable and may require an emergency take-back to the operating room. This affects outcomes and costs of care.[citation needed]
- Most studies researching clopidogrel do not compare patients on clopidogrel to patients taking placebo; rather, clopidogrel use is compared to aspirin use. Thus, attributing side effects directly to clopidogrel is difficult. Other side effects may include:
- Other gastrointestinal side effects
- Upper GI discomfort (27% vs 29% in patients taking aspirin alone)
- Gastric or duodenal ulcer, gastritis
- Diarrhea (4.5% of patients in the CAPRIE trial)[17]
- Rash (6% overall, 0.33% severe)[18]
- Respiratory (infrequent)
- Upper respiratory infections, rhinitis, shortness of breath, cough
- Cardiovascular
- chest pain
- edema (generalized swelling)
- Thrombocytopenia (reduction of platelets, 0.2% severe cases as compared to 0.1% under aspirin)
 | |
| Systematic (IUPAC) name | |
|---|---|
| (+)-(S)-methyl 2-(2-chlorophenyl)-2-(6,7-dihydrothieno[3,2-c]pyridin-5(4H)-yl)acetate | |
| Clinical data | |
| Trade names | Plavix |
| AHFS/Drugs.com | monograph |
| MedlinePlus | a601040 |
| Licence data | US FDA:link |
| Pregnancy cat. | B1 (AU) B (US) |
| Legal status | Prescription Only (S4) (AU)POM (UK) ℞-only (US) |
| Routes | Oral |
| Pharmacokinetic data | |
| Bioavailability | >50% |
| Protein binding | 94–98% |
| Metabolism | Hepatic |
| Half-life | 7–8 hours (inactive metabolite) |
| Excretion | 50% renal 46% biliary |
| Identifiers | |
| CAS number | 113665-84-2 |
| ATC code | B01AC04 |
| PubChem | CID 60606 |
| DrugBank | DB00758 |
| ChemSpider | 54632 |
| UNII | A74586SNO7 |
| KEGG | D07729 |
| ChEBI | CHEBI:37941 |
| ChEMBL | CHEMBL1771 |
| Chemical data | |
| Formula | C16H16ClNO2S |
| Mol. mass | 321.82 g/mol |
Interactions
Clopidogrel interacts with: phenytoin (Dilantin), tamoxifen (Nolvadex), tolbutamide (Orinase), torsemide (Demadex), fluvastatin (Lescol), a blood thinner such as warfarin(Coumadin), heparin, ardeparin (Normiflo), dalteparin (Fragmin), danaparoid (Orgaran), enoxaparin (Lovenox), or tinzaparin (Innohep), anistreplase (Eminase), dipyridamole(Persantine), streptokinase (Kabikinase, Streptase), ticlopidine (Ticlid), and urokinase (Abbokinase).
In November 2009, the FDA announced that clopidogrel should be used with caution in patients using the proton pump inhibitors omeprazole or esomeprazole,[19][20] butpantoprazole appears to be safe.[21] The newer antiplatelet agent prasugrel has minimal interaction with (es)omeprazole, hence might be a better antiplatelet agent (if no other contraindications are present) in patients who are on these proton pump inhibitors.[22]
Pharmacology
Clopidogrel is a prodrug, which requires CYP2C19 for its activation.[23] It acts on the ADP receptor on platelet cell membranes. The drug specifically and irreversibly inhibits theP2Y12 subtype of ADP receptor, which is important in activation of platelets and eventual cross-linking by the protein fibrin.[24] Platelet inhibition can be demonstrated two hours after a single dose of oral clopidogrel, but the onset of action is slow, so a loading dose of either 600 or 300 mg is administered when a rapid effect is needed.[25]
Pharmacokinetics and metabolism
After repeated oral doses of 75 mg of clopidogrel (base), plasma concentrations of the parent compound, which has no platelet-inhibiting effect, are very low and, in general, are below the quantification limit (0.258 µg/l) beyond two hours after dosing.[citation needed]
Clopidogrel is activated in the liver by cytochrome P450 enzymes, including CYP2C19. Due to opening of the thiophene ring, the chemical structure of the active metabolite has three sites that are stereochemically relevant, making a total of eight possible isomers. These are: a stereocentre at C4 (attached to the —SH thiol group), a double bond at C3—C16, and the original stereocentre at C7. Only one of the eight structures is an active antiplatelet drug. This has the following configuration: Z configuration at the C3—C16 double bond, the original S configuration at C7,[26] and, although the stereocentre at C4 cannot be directly determined, as the thiol group is too reactive, work with the active metabolite of the related drug prasugrel suggests the R-configuration of the C4 group is critical for P2Y12 and platelet-inhibitory activity.[citation needed]
The active metabolite has an elimination half-life of about 0.5 to 1.0 h, and acts by forming a disulfide bridge with the platelet ADP receptor. Patients with a variant allele of CYP2C19 are 1.5 to 3.5 times more likely to die or have complications than patients with the high-functioning allele.[27][28][29]
Following an oral dose of 14C-labeled clopidogrel in humans, about 50% was excreted in the urine and 46% in the feces in the five days after dosing.
- Effect of food: Administration of clopidogrel bisulfate with meals did not significantly modify the bioavailability of clopidogrel as assessed by the pharmacokinetics of the main circulating metabolite.
- Absorption and distribution: Clopidogrel is rapidly absorbed after oral administration of repeated doses of 75-milligram clopidogrel (base), with peak plasma levels (about 3 mg/l) of the main circulating metabolite occurring around one hour after dosing. The pharmacokinetics of the main circulating metabolite are linear (plasma concentrations increased in proportion to dose) in the dose range of 50 to 150 mg of clopidogrel. Absorption is at least 50% based on urinary excretion of clopidogrel-related metabolites.
Clopidogrel and the main circulating metabolite bind reversibly in vitro to human plasma proteins (98% and 94%, respectively). The binding is not saturable in vitro up to a concentration of 110 μg/ml.
- Metabolism and elimination: In vitro and in vivo, clopidogrel undergoes rapid hydrolysis into its carboxylic acid derivative. In plasma and urine, the glucuronide of the carboxylic acid derivative is also observed.
In March 2010, the U.S. FDA added a boxed warning to Plavix alerting that the drug can be less effective in people unable to metabolize the drug to convert it to its active form.[30][31]
Pharmacogenetics
CYP2C19 is an important drug-metabolizing enzyme that catalyzes the biotransformation of many clinically useful drugs, including antidepressants, barbiturates, proton pump inhibitors, and antimalarial and antitumor drugs. Clopidogrel is one of the drugs metabolized by this enzyme.
Several recent landmark studies have proven the importance of 2C19 genotyping in treatment using clopidogrel. In March 2010, the FDA put a black box warning on Plavix to make patients and healthcare providers aware that CYP2C19-poor metabolizers, representing up to 14% of patients, are at high risk of treatment failure and that testing is available.[30] Patients with variants in cytochrome P-450 2C19 (CYP2C19) have lower levels of the active metabolite of clopidogrel, less inhibition of platelets, and a 3.58-times greater risk for major adverse cardiovascular events such as death, heart attack, and stroke; the risk was greatest in CYP2C19 poor metabolizers.[32]
Marketing and litigation
Plavix is marketed worldwide in nearly 110 countries, with sales of US$6.6 billion in 2009.[33] It had been the second-top-selling drug in the world for a few years as of 2007[34] and was still growing by over 20% in 2007. U.S. sales were US$3.8 billion in 2008.[35]
In 2006, generic clopidogrel was briefly marketed by Apotex, a Canadian generic pharmaceutical company before a court order halted further production until resolution of a patent infringement case brought by Bristol-Myers Squibb.[36] The court ruled that Bristol-Myers Squibb's patent was valid and provided protection until November 2011.[37] The FDA extended the patent protection of clopidogrel by six months, giving exclusivity that would expire on May 17, 2012.[38] The FDA approved generic versions of Plavix on May 17, 2012.[39]
In June 2009, the European Medicines Agency gave authorisation to six generic versions of clopidogrel bisulfate, and the drug is now available in several European countries, including the United Kingdom.[40]
Generic clopidogrel is produced by several pharmaceutical companies in India and elsewhere, and often sold under its INN. It is marketed by Cipla under the trade name Clopivas, by Sun Pharmaceuticals as Clopilet, by Ranbaxy Laboratories as Ceruvin, as Clavix by Intas Pharmaceuticals, and as Deplatt by Torrent Pharmaceuticals. In India, it is sold as Clopivas AP by Cipla, Clopigrel A by USV, and Clopitab A by Lupin(mixed with aspirin).[citation needed]
Generic clopidogrel is produced in Slovenia (European Union) under the trade names Zyllt, Kardogrel and Clopidogrel Krka by Krka d.d., Novo Mesto.[41]
Another generic clopidogrel is produced for Julphar brand (Gulf Pharmaceutical Industries) UAE (GCC) as Lavigard, and by EGIS Pharmaceuticals PLC, Budapest, Hungary, as Clopidogrel.
Dosage forms
Clopidogrel is marketed as clopidogrel bisulfate (clopidogrel hydrogen sulfate), most commonly under the trade name Plavix, as 75- and 300-mg oral tablets.[42]
Use in cat
Clopidogrel has also been shown to be effective at decreasing platelet aggregation in cats, so its use in prevention of feline aortic thromboembolism has been advocated.[43]
Racemic Synthesis 1
Racemic Synthesis 2
Asymmetric Synthesis 1
- CSA means Camphorsulfonic acid (c.f. Trimetaphan camsilate).
Asymmetric Synthesis 2
References
- ^ Topol, Eric J; Schork, Nicholas J (January 2011). "Catapulting clopidogrel pharmacogenomics forward". Nature Medicine 17 (1): 40–41. doi:10.1038/nm0111-40. PMID 21217678.
- ^ O'Gara PT, Kushner FG, Ascheim DD, et al. (January 2013). "2013 ACCF/AHA guideline for the management of ST-elevation myocardial infarction: a report of the American College of Cardiology Foundation/American Heart Association Task Force on Practice Guidelines". J. Am. Coll. Cardiol. 61 (4): e78–140.doi:10.1016/j.jacc.2012.11.019. PMID 23256914.
- ^ Jneid H, Anderson JL, Wright RS, et al. (August 2012). "2012 ACCF/AHA focused update of the guideline for the management of patients with unstable angina/Non-ST-elevation myocardial infarction (updating the 2007 guideline and replacing the 2011 focused update): a report of the American College of Cardiology Foundation/American Heart Association Task Force on practice guidelines". Circulation 126 (7): 875–910.doi:10.1161/CIR.0b013e318256f1e0. PMID 22800849.
- ^ Fihn SD, Gardin JM, Abrams J, et al. (December 2012). "2012 ACCF/AHA/ACP/AATS/PCNA/SCAI/STS guideline for the diagnosis and management of patients with stable ischemic heart disease: executive summary: a report of the American College of Cardiology Foundation/American Heart Association task force on practice guidelines, and the American College of Physicians, American Association for Thoracic Surgery, Preventive Cardiovascular Nurses Association, Society for Cardiovascular Angiography and Interventions, and Society of Thoracic Surgeons".Circulation 126 (25): 3097–137. doi:10.1161/CIR.0b013e3182776f83.PMID 23166210.
- ^ Rossi S, editor. Australian Medicines Handbook 2006. Adelaide: Australian Medicines Handbook; 2006. ISBN 0-9757919-2-3
- ^ Michael D Randall; Karen E Neil (2004). Disease management. 2nd ed. London: Pharmaceutical Press. 159.
- ^ Gagne, JJ; Bykov, K; Choudhry, NK; Toomey, TJ; Connolly, JG; Avorn, J (Sep 17, 2013). "Effect of smoking on comparative efficacy of antiplatelet agents: systematic review, meta-analysis, and indirect comparison.". BMJ (Clinical research ed.) 347: f5307.doi:10.1136/bmj.f5307. PMID 24046285.
- ^ Chan FK; Ching JY; Hung LC; et al. (2005). "Clopidogrel versus aspirin and esomeprazole to prevent recurrent ulcer bleeding". N. Engl. J. Med. (PDF) 352 (3): 238–44. doi:10.1056/NEJMoa042087. PMID 15659723.
- ^ Mistry SD, Trivedi HR, Parmar DM, Dalvi PS, Jiyo C. (2011). "Impact of proton pump inhibitors on efficacy of clopidogrel: Review of evidence". Indian Journal of Pharmacology 43 (2): 183–6. doi:10.4103/0253-7613.77360. PMC 3081459.PMID 21572655.
- ^ Ho PM, Maddox TM, Wang L, Fihn SD, Jesse RL, Peterson ED, Rumsfeld JS. (2009). "Risk of adverse outcomes associated with concomitant use of clopidogrel and proton pump inhibitors following acute coronary syndrome". Journal of the American Medical Association 301 (9): 937–44. doi:10.1001/jama.2009.261. PMID 19258584.
- ^ Stockl, KM; Le L; Zakharyan A; et al. (April 2010). "Risk of rehospitalization for patients using clopidogrel with a proton pump inhibitor" (PDF). Arch Intern Med 170 (8): 704–10. doi:10.1001/archinternmed.2010.34. ISSN 1538-3679. PMID 20421557.
- ^ Wathion, Noël. "Public statement on possible interaction between clopidogrel and proton pump inhibitors". Retrieved 31 March 2011.
- ^ Hughes, Sue. "EMEA issues warning on possible clopidogrel-PPI interaction, but is there really a problem?". Retrieved 31 March 2011.
- ^ Zakarija A; Bandarenko N; Pandey DK; et al. (2004). "Clopidogrel-Associated TTP An Update of Pharmacovigilance Efforts Conducted by Independent Researchers, Pharmaceutical Suppliers, and the Food and Drug Administration". Stroke 35 (2): 533–8. doi:10.1161/01.STR.0000109253.66918.5E. PMID 14707231.
- ^ Plavix prescribing information. Bristol-Myers Squibb / Sanofi.
- ^ Diener HC; Bogousslavsky J; Brass LM; et al. (2004). "Aspirin and clopidogrel compared with clopidogrel alone after recent ischaemic stroke or transient ischaemic attack in high-risk patients (MATCH): randomised, double-blind, placebo-controlled trial".The Lancet 364 (9431): 331–7. doi:10.1016/S0140-6736(04)16721-4.PMID 15276392.
- ^ Harker LA, Boissel JP, Pilgrim AJ, Gent M (October 1999). "Comparative safety and tolerability of clopidogrel and aspirin: results from CAPRIE. CAPRIE Steering Committee and Investigators. Clopidogrel versus aspirin in patients at risk of ischaemic events.".Drug Saf. 21 (4): 325–35. doi:10.2165/00002018-199921040-00007. PMID 10514023.
- ^ none listed (November 1996). "A randomised, blinded, trial of clopidogrel versus aspirin in patients at risk of ischaemic events (CAPRIE). CAPRIE Steering Committee". The Lancet 16 (9038): 1329–39. PMID 8918275.
- ^ DeNoon, Daniel J. "FDA Warns Plavix Patients of Drug Interactions", WebMD, 2009-11-19. Retrieved 2009-11-23.
- ^ "Public Health Advisory: Updated Safety Information about a drug interaction between Clopidogrel Bisulfate (marketed as Plavix) and Omeprazole (marketed as Prilosec and Prilosec OTC)". Food and Drug Administration (FDA). November 17, 2009. Archived from the original on 2009-12-29. Retrieved March 13, 2010.
- ^ Wedemeyer, R. S.; Blume, H. (2014). "Pharmacokinetic Drug Interaction Profiles of Proton Pump Inhibitors: An Update". Drug Safety 37 (4): 201. doi:10.1007/s40264-014-0144-0.
- ^ John, Jinu; Koshy (2012). "Current Oral Antiplatelets: Focus Update on Prasugrel".Journal of American Board of Family Medicine 25 (3): 343–349.doi:10.3122/jabfm.2012.03.100270.
- ^ http://www.ncbi.nlm.nih.gov/pubmed/23046071
- ^ Savi, P; Zachayus JL; Delesque-Touchard N; et al. (July 2006). "The active metabolite of Clopidogrel disrupts P2Y12 receptor oligomers and partitions them out of lipid rafts". Proceedings of the National Academy of Sciences of the USA 103 (29): 11069–11074. doi:10.1073/pnas.0510446103. PMC 1635153. PMID 16835302.
- ^ Clopidogrel Multum Consumer Information
- ^ a b Pereillo JM, Maftouh M, Andrieu A, Uzabiaga MF, Fedeli O, Savi P, Pascal M, Herbert JM, Maffrand JP, Picard C (2002). "Structure and stereochemistry of the active metabolite of clopidogrel". Drug Metab. Dispos. 30 (11): 1288–95.doi:10.1124/dmd.30.11.1288. PMID 12386137.
- ^ Mega JL; Close, SL; Wiviott, SD; Shen, L; Hockett, RD; Brandt, JT; Walker, JR; Antman, EM et al. (January 2009). "Cytochrome p-450 polymorphisms and response to clopidogrel". The New England Journal of Medicine (PDF) 360 (4): 354–62.doi:10.1056/NEJMoa0809171. PMID 19106084.
- ^ Simon T; Verstuyft, C; Mary-Krause, M; Quteineh, L; Drouet, E; Méneveau, N; Steg, PG; Ferrières, J et al. (January 2009). "Genetic Determinants of Response to Clopidogrel and Cardiovascular Events". The New England Journal of Medicine (PDF) 360 (4): 363–75. doi:10.1056/NEJMoa0808227. PMID 19106083.
- ^ Collet, J; Hulot, JS; Pena, A; Villard, E; Esteve, JB; Silvain, J; Payot, L; Brugier, D et al. (January 2009). "Cytochrome P450 2C19 polymorphism in young patients treated with clopidogrel after myocardial infarction: a cohort study". The Lancet 373 (9660): 309–17.doi:10.1016/S0140-6736(08)61845-0. PMID 19108880.
- ^ a b "FDA Announces New Boxed Warning on Plavix: Alerts patients, health care professionals to potential for reduced effectiveness" (Press release). Food and Drug Administration (FDA). March 12, 2010. Retrieved March 13, 2010.
- ^ "FDA Drug Safety Communication: Reduced effectiveness of Plavix (clopidogrel) in patients who are poor metabolizers of the drug". Food and Drug Administration (FDA). March 12, 2010. Retrieved March 13, 2010.
- ^ PGxNews.Org (June 2009). "FDA updates Plavix label with PGx data". PGxNews.Org. Retrieved 2009-06-13.[dead link]
- ^ "New products and markets fuel growth in 2005". IMS Health. Retrieved 2009-03-02.[dead link]
- ^ "Top Ten Global Products - 2007" (PDF). IMS Health. 2008-02-26. Retrieved 2009-03-02.
- ^ "Details for Plavix".[verification needed]
- ^ "Preliminary Injunction Against Apotex Upheld on Appeal" (Press release). Bristol-Myers Squibb. December 8, 2006. Retrieved 2010-03-14.
- ^ "U.S. judge upholds Bristol, Sanofi patent on Plavix". Reuters. June 19, 2007. Retrieved 2007-09-05.
- ^ "FDA Gives Plavix a 6 Month Extension-Patent Now Expires on May 17, 2012". January 26, 2011. Retrieved 2012-01-12.
- ^ "FDA approves generic versions of blood thinner Plavix" (Press release). Food and Drug Administration (FDA). May 17, 2012.
- ^ Generic Clopidogrel http://www.genericclopidogrel.com/generic-clopidogrel/
- ^ Krka official website http://www.krka.biz/en/pharmaceuticals-and-other-products/prescription-pharmaceuticals/?searchTerm=clopidogrel
- ^ Joint Royal Colleges Ambulance liaison committee Clinical Practise Guideines Guidelines
- ^ Hogan DF, Andrews DA, Green HW, et al. (2004). "Antiplatelet effects and pharmacodynamics of clopidogrel in cats". J Am Vet Med Assoc 225 (9): 1406–1411.
External links
- Bristol-Myers Squibb official website Bristol-Myers Squibb/Sanofi
- Plavix prescribing information Bristol-Myers Squibb/Sanofi
- Plavix, Aspirin and Stents : Patients' Forum : Angioplasty.Org
- Drug promises end to migraine misery
- U.S. National Library of Medicine: Drug Information Portal - Clopidogrel
- Krka official website
Racemic Synthesis 1
Racemic Synthesis 2
Asymmetric Synthesis 1
- CSA means Camphorsulfonic acid (c.f. Trimetaphan camsilate).






